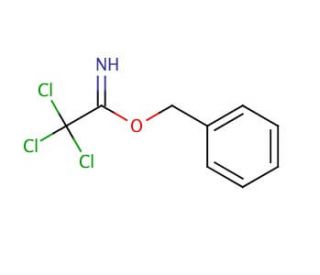
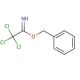
Benzyl 2,2,2-trichloroacetimidate (CAS 81927-55-1)
QUICK LINKS
Benzyl 2,2,2-trichloroacetimidate functions as a reagent in organic synthesis. It acts as a protecting group for amines, allowing for selective deprotection under mild conditions. Benzyl 2,2,2-trichloroacetimidate′s mechanism of action involves the reaction of the trichloroacetimidate group with the amine, forming a stable imidate intermediate. This intermediate can then undergo further reactions, while the benzyl group protects the amine from unwanted reactions. In this way, Benzyl 2,2,2-trichloroacetimidate plays a role in the controlled manipulation of amine functionality in the synthesis of complex organic molecules. Its ability to selectively protect and deprotect amines may be useful in the construction of diverse chemical structures.
Benzyl 2,2,2-trichloroacetimidate (CAS 81927-55-1) References
- Synthesis and characterization of poly(butylene succinate-co-butylene malate): a new biodegradable copolyester bearing hydroxyl pendant groups. | Zhang, S., et al. 2003. Biomacromolecules. 4: 437-45. PMID: 12625743
- Design and synthesis of paromomycin-related heterocycle-substituted aminoglycoside mimetics based on a mass spectrometry RNA-binding assay. | Ding, Y., et al. 2003. Angew Chem Int Ed Engl. 42: 3409-12. PMID: 12888972
- Silanediol peptidomimetics. Evaluation of four diastereomeric ACE inhibitors. | Kim, J. and Sieburth, SM. 2004. Bioorg Med Chem Lett. 14: 2853-6. PMID: 15125946
- Alkene-alkyne coupling as a linchpin: an efficient and convergent synthesis of amphidinolide P. | Trost, BM. and Papillon, JP. 2004. J Am Chem Soc. 126: 13618-9. PMID: 15493910
- A comparative study of the influence of some protecting groups on the reactivity of D-glucosamine acceptors with a galactofuranosyl donor. | Bohn, ML., et al. 2006. Carbohydr Res. 341: 1096-104. PMID: 16630599
- Synthesis of rhamnogalacturonan I fragments by a modular design principle. | Nemati, N., et al. 2008. Carbohydr Res. 343: 1730-42. PMID: 18378221
- Total synthesis of N-acetylglucosamine-1,6-anhydro-N-acetylmuramylpentapeptide and evaluation of its turnover by AmpD from Escherichia coli. | Hesek, D., et al. 2009. J Am Chem Soc. 131: 5187-93. PMID: 19309146
- A novel acid-catalyzed O-benzylating reagent with the smallest unit of imidate structure. | Yamada, K., et al. 2012. Org Lett. 14: 5026-9. PMID: 22994426
- Environmentally benign process for the preparation of antimicrobial α-methylene-β-hydroxy-γ-butyrolactone (tulipalin B) from tulip biomass. | Nomura, T., et al. 2015. Biosci Biotechnol Biochem. 79: 25-35. PMID: 25126881
- Design of selective COX-2 inhibitors in the (aza)indazole series. Chemistry, in vitro studies, radiochemistry and evaluations in rats of a [18F] PET tracer. | Elie, J., et al. 2019. J Enzyme Inhib Med Chem. 34: 1-7. PMID: 30362376
- Ester Formation via Symbiotic Activation Utilizing Trichloroacetimidate Electrophiles. | Mahajani, NS., et al. 2019. J Org Chem. 84: 7871-7882. PMID: 31117564
- Extended scaffold glucuronides: en route to the universal synthesis of O-aryl glucuronide prodrugs. | Walther, R., et al. 2019. Org Biomol Chem. 17: 6970-6974. PMID: 31290904
- Benzyl trichloroacetimidates as derivatizing agents for phosphonic acids related to nerve agents by EI-GC-MS during OPCW proficiency test scenarios. | Subramanian, A., et al. 2022. Sci Rep. 12: 21299. PMID: 36494565
- Activation of thioglycosides under mild alkylation conditions. | Forsythe, N., et al. 2023. Carbohydr Res. 531: 108872. PMID: 37348387
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Benzyl 2,2,2-trichloroacetimidate, 5 g | sc-252423 | 5 g | $41.00 |
