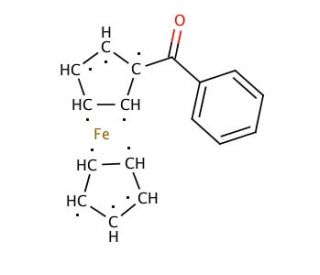
Molecular structure of Benzoylferrocene, CAS Number: 1272-44-2
Benzoylferrocene (CAS 1272-44-2)
Alternate Names:
(Benzoylcyclopentadienyl)cyclopentadienyliron
CAS Number:
1272-44-2
Molecular Weight:
290.14
Molecular Formula:
C17H14FeO
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Benzoylferrocene is a radical initiator in various polymerization reactions. Benzoylferrocene′s mechanism of action involves the generation of free radicals through thermal or photochemical processes, which then initiate the polymerization of monomers. Benzoylferrocene plays a role in the initiation of polymerization reactions by facilitating the formation of active species that can react with monomers to form polymers. Benzoylferrocene′s mechanism of action involves the cleavage of the peroxide bond, leading to the generation of alkoxyl and benzoyloxy radicals that can initiate polymerization.
Benzoylferrocene (CAS 1272-44-2) References
- Selective hydrogenation of benzophenones to benzhydrols. Asymmetric synthesis of unsymmetrical diarylmethanols. | Ohkuma, T., et al. 2000. Org Lett. 2: 659-62. PMID: 10814403
- Synthesis of ferrocenyl alkenes, dienes, and enynes via samarium diiodide promoted tandem addition and dehydration of ferrocenyl carbonyls with halides. | Jong, SJ. and Fang, JM. 2001. J Org Chem. 66: 3533-7. PMID: 11348141
- Characterization of the low-energy electronic excited States of benzoyl-substituted ferrocenes. | Ding, W., et al. 2003. Inorg Chem. 42: 1532-7. PMID: 12611520
- Photoreactivity and photopolymerization of silicon-bridged [1]ferrocenophanes in the presence of terpyridine initiators: unprecedented cleavage of both iron-cyclopentadienyl bonds in the presence of chlorosilanes. | Chan, WY., et al. 2007. Chemistry. 13: 8867-76. PMID: 17654755
- Synthesis of hydroxyferrocifen and its abilities to protect DNA and to scavenge radicals. | Zhao, F., et al. 2011. J Biol Inorg Chem. 16: 1169-76. PMID: 21701897
- Assessing the reactivity of sodium zincate [(TMEDA)Na(TMP)Zn(t)Bu2] towards benzoylferrocene: deprotonative metalation vs. alkylation reactions. | Hevia, E., et al. 2012. Dalton Trans. 41: 98-103. PMID: 22024728
- Electrochemical sensor for selective determination of N-acetylcysteine in the presence of folic acid using a modified carbon nanotube paste electrode. | Beitollahi, H., et al. 2013. Mater Sci Eng C Mater Biol Appl. 33: 1078-84. PMID: 23827545
- Thermodynamic properties of benzoylferrocene and 1,1′-dibenzoylferrocene | M. Pelino, R. Gigli, M. Tomassetti. 1983. Thermochimica Acta. 61: 301-305.
- Synthesis and mercuration of anils of benzoylferrocene | Shou Quan Huo, Yang Jie Wu ∗, Xi An Mao, Han Zhen Yuan. 1994. Tetrahedron. 50: 10467-10476.
- Synthesis and characterization of novel heteroannular benzoylferrocene polysiloxane monomers, oligomers, and polymers | JonathanR. Sargent, WilliamP. Weber. 1999. Polymer. 40: 3795-3802.
- Electrocatalytic measurement of methionine concentration with a carbon nanotube paste electrode modified with benzoylferrocene | Hadi BEITOLLAHI a, Alireza MOHADESI b, Farzaneh GHORBANI b, Hassan KARIMI MALEH c, Mehdi BAGHAYERI d, Rahman HOSSEINZADEH e. 2013. Chinese Journal of Catalysis. 34: 1333-1338.
- Syntheses, third-order optical nonlinearity and DFT studies on benzoylferrocene derivatives | Jianhong Jia, Yanhong Cui, Liang Han, Weijian Sheng, Yujin Li, Jianrong Gao. 2014. Dyes and Pigments. 104: 137-145.
- Chemical curing of alkyd resin catalyzed by benzoylferrocene: Performance, kinetics, and thickness effects | Jan Honzíček, Jaromír Vinklárek. 2018. Journal of Applied Polymer Science. 135.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Benzoylferrocene, 5 g | sc-227329 | 5 g | $131.00 |
