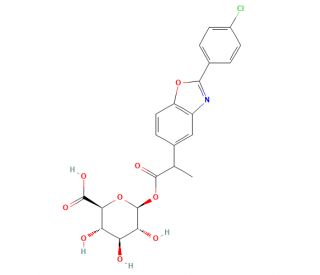
Benoxaprofen Glucuronide (CAS 67472-42-8)
QUICK LINKS
Benoxaprofen Glucuronide, a metabolite of the parent compound benoxaprofen, plays a significant role in understanding the pharmacokinetics and metabolic pathways of nonsteroidal anti-inflammatory compounds in research environments. As a conjugate where benoxaprofen is linked to glucuronic acid, this compound is primarily formed in the liver through the enzymatic process of glucuronidation, a key phase II drug metabolism reaction which enhances the solubility of hydrophobic molecules and facilitates their excretion. In scientific studies, Benoxaprofen Glucuronide serves as a crucial marker to investigate the efficiency and pathways of glucuronidation reactions in different species, thereby providing insights into interspecies differences and potential xenobiotic interactions. Researchers leverage this compound to explore the dynamics of drug clearance and to assess the impact of metabolic stability on the excretion profiles of pharmaceutical substances. By studying Benoxaprofen Glucuronide, scientists can better understand how modifications in molecular structure influence metabolic pathways, which is essential for the design and development of safer and more effective molecules. This research is invaluable in the field of toxicology and pharmacology, where understanding the metabolic fate of chemicals is crucial for predicting their behavior in biological systems.
Benoxaprofen Glucuronide (CAS 67472-42-8) References
- Effect of selective phase II enzyme inducers on glucuronidation of benoxaprofen in rats. | Dong, JQ., et al. 1999. Drug Metab Dispos. 27: 1423-8. PMID: 10570023
- Role of benoxaprofen and flunoxaprofen acyl glucuronides in covalent binding to rat plasma and liver proteins in vivo. | Dong, JQ., et al. 2005. Biochem Pharmacol. 70: 937-48. PMID: 16046212
- In vitro enantioselective glucuronidation of fenoprofen. | Volland, C. and Benet, LZ. 1991. Pharmacology. 43: 53-60. PMID: 1775510
- Glucuronidation and covalent protein binding of benoxaprofen and flunoxaprofen in sandwich-cultured rat and human hepatocytes. | Dong, JQ. and Smith, PC. 2009. Drug Metab Dispos. 37: 2314-22. PMID: 19773537
- Effects of clofibric acid on the biliary excretion of benoxaprofen glucuronide and taurine conjugate in rats. | Okada, K., et al. 2011. Pharmazie. 66: 777-83. PMID: 22026160
- [Analysis of benoxaprofen glucuronide in human plasma and urine by gas chromatography]. | Okamoto, J., et al. 1983. Yakugaku Zasshi. 103: 54-61. PMID: 6602873
- Pharmacology, clinical efficacy, and adverse effects of the nonsteroidal anti-inflammatory agent benoxaprofen. | Dahl, SL. and Ward, JR. 1982. Pharmacotherapy. 2: 354-66. PMID: 6762531
- Mechanisms for covalent binding of benoxaprofen glucuronide to human serum albumin. Studies By tandem mass spectrometry. | Qiu, Y., et al. 1998. Drug Metab Dispos. 26: 246-56. PMID: 9492388
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Benoxaprofen Glucuronide, 0.5 mg | sc-503280 | 0.5 mg | $463.00 | |||
Benoxaprofen Glucuronide, 5 mg | sc-503280A | 5 mg | $3334.00 |
