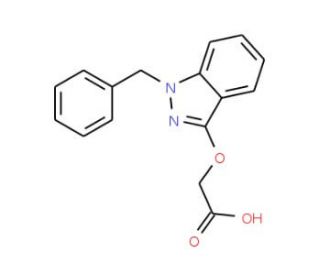
Molecular structure of Bendazac, CAS Number: 20187-55-7
Bendazac (CAS 20187-55-7)
Alternate Names:
1-Benzylindazole-3-oxyacetic acid; Bendazolic acid; Zildasac; Versus
Application:
Bendazac is inhibits the denaturing of proteins
CAS Number:
20187-55-7
Molecular Weight:
282.29
Molecular Formula:
C16H14N2O3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Bendazac inhibits the denaturing of proteins. Bendazac (or bendazolic acid) is a non-steroidal anti-inflammatory drug used for joint and muscular pain. Bendazac has been used in studies related to potential treatment and prevention of ischemia and reperfusion syndromes, and was found to inhibit only reversible and irreversible xanthine oxidase (mixed non-competitive-competitive).
Bendazac (CAS 20187-55-7) References
- Kinetics of the inhibition of xanthine dehydrogenase and of the reversible and irreversible forms of xanthine oxidase by silibinin and bendazac. | Silva, MP., et al. 1996. Environ Toxicol Pharmacol. 1: 279-84. PMID: 21781693
- Bendazac lysine. A review of its pharmacological properties and therapeutic potential in the management of cataracts. | Balfour, JA. and Clissold, SP. 1990. Drugs. 39: 575-96. PMID: 2190795
- Antioxidant characteristics of some potential anticataract agents. Studies of aspirin, paracetamol, and bendazac provide support for an oxidative component of cataract. | Woollard, AC., et al. 1990. Free Radic Biol Med. 9: 299-305. PMID: 2283086
- The pharmacokinetics of bendazac-lysine and 5-hydroxybendazac, its main metabolite, in patients with hepatic cirrhosis. | Rovei, V., et al. 1988. Eur J Clin Pharmacol. 35: 391-6. PMID: 3197747
- The major metabolite of bendazac inhibits the glycosylation of soluble lens proteins: a possible mechanism for a delay in cataractogenesis. | Lewis, BS. and Harding, JJ. 1988. Exp Eye Res. 47: 217-25. PMID: 3409993
- Bendazac and benzydamine for treatment of cataract: individualized therapy by the 'BLOA test'. | Testa, M., et al. 1986. J Ocul Pharmacol. 2: 251-66. PMID: 3503111
- Engineering a multi-target therapy nanoplatform against tumor growth and metastasis via a novel NSAID-Pt(IV) prodrug. | Zhang, D., et al. 2022. Chem Commun (Camb). 58: 3803-3806. PMID: 35229844
- Pharmacokinetics of bendazac-lysine and 5-hydroxybendazac in patients with renal insufficiency. | Rovei, V., et al. 1987. Eur J Clin Pharmacol. 33: 303-10. PMID: 3691618
- Exploring Synthetic Strategies for 1H-Indazoles and Their N-Oxides: Electrochemical Synthesis of 1H-Indazole N-Oxides and Their Divergent C-H Functionalizations. | Arepally, S., et al. 2023. Angew Chem Int Ed Engl. 62: e202303460. PMID: 37086084
- Ocular absorption and distribution of bendazac after topical administration to rabbits with different vehicles. | Romanelli, L., et al. 1994. Life Sci. 54: 877-85. PMID: 8139376
- Bendazac decreases in vitro glycation of human lens crystallins. Decrease of in vitro protein glycation by bendazac. | Marques, C., et al. 1995. Doc Ophthalmol. 90: 395-404. PMID: 8620822
Inhibitor of:
Xanthine Oxidase.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Bendazac, 5 g | sc-337553 | 5 g | $180.00 |
