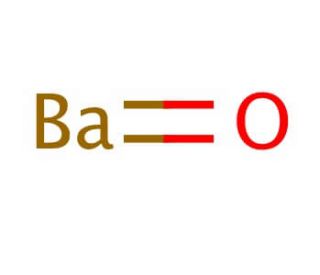
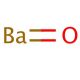
Barium oxide (CAS 1304-28-5)
QUICK LINKS
Barium oxide, a versatile compound in scientific research, is distinguished by its unique mechanisms of action and widespread applications across various disciplines. In research, barium oxide serves as a crucial component in materials science and catalysis. Its mechanism of action primarily revolves around its high reactivity and ability to function as a strong base. Barium oxide readily reacts with water to form barium hydroxide, releasing hydroxide ions in the process. This alkaline property enables its utility in diverse reactions, including the synthesis of perovskite-type materials, where barium oxide acts as a precursor in the formation of complex oxides with desirable properties like ferroelectricity and magnetism. Moreover, barium oxide finds application as a catalyst support or active catalyst in numerous chemical transformations, such as the production of fine chemicals and petrochemicals. Its catalytic activity stems from its ability to participate in redox reactions, acid-base catalysis, and surface-mediated processes. Additionally, in environmental research, barium oxide plays a role in waste remediation and pollution control, owing to its capability to adsorb and neutralize acidic pollutants. Furthermore, in energy research, barium oxide-based materials are investigated for their potential in energy storage and conversion devices, including batteries and fuel cells. Overall, the multifaceted properties and reactivity of barium oxide make it an indispensable tool in advancing scientific knowledge and technological innovation across various research domains.
Barium oxide (CAS 1304-28-5) References
- Strain-induced ferroelectricity in simple rocksalt binary oxides. | Bousquet, E., et al. 2010. Phys Rev Lett. 104: 037601. PMID: 20366683
- Synthesis of protected glucose derivatives from levoglucosan by development of common carbohydrate protecting group reactions under continuous flow conditions. | Marion, KC., et al. 2018. Carbohydr Res. 468: 23-29. PMID: 30121415
- Investigation of bismuth borate glass system modified with barium for structural and gamma-ray shielding properties. | Kaur, P., et al. 2019. Spectrochim Acta A Mol Biomol Spectrosc. 206: 367-377. PMID: 30157444
- Pharmacological application of barium containing bioactive glass in gastro-duodenal ulcers. | Paliwal, P., et al. 2018. Mater Sci Eng C Mater Biol Appl. 92: 424-434. PMID: 30184768
- Exploring glycosyl sulphates as donors for chemical glycosylation. | Imperio, D., et al. 2021. Org Biomol Chem. 19: 4930-4936. PMID: 33982734
- Barium Oxide Doped Magnesium Silicate Nanopowders for Bone Fracture Healing: Preparation, Characterization, Antibacterial and In Vivo Animal Studies. | Mabrouk, M., et al. 2022. Pharmaceutics. 14: PMID: 36015208
- Coating of polyester fabrics with micro-particles of Bi2O3 and BaO for ionization ray shielding. | Mehrjardi, AZ., et al. 2023. Appl Radiat Isot. 192: 110573. PMID: 36473317
- Design of the Lanthanum hexaboride based plasma source for the large plasma device at UCLA. | Qian, Y., et al. 2023. Rev Sci Instrum. 94: PMID: 38065131
- Deep insight into physical properties of carboxymethyl cellulose-barium oxide nanocomposites. | Bashal, AH., et al. 2024. Int J Biol Macromol. 269: 131935. PMID: 38723542
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Barium oxide, 500 g | sc-210860 | 500 g | $79.00 |
