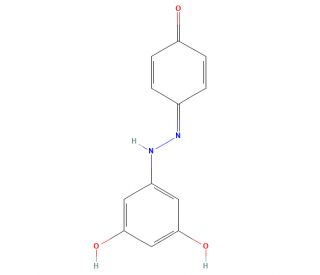
Azo-Resveratrol (CAS 1393556-48-3)
QUICK LINKS
Azo-Resveratrol, a versatile chemical compound, finds extensive use in various scientific research applications. As a diol, it features two hydroxyl groups attached to a single carbon atom, rendering it a compound of great interest for its distinctive properties and potential across multiple fields. It serves as a key component in the synthesis of polymers, dyes, and other materials. Furthermore, it plays a significant role in the development of pharmaceuticals, including anti-cancer and anti-inflammatory agents. Its utility extends to the study of protein structure and function, as well as the exploration of enzyme inhibitors. Although the exact mechanism of action of Azo-Resveratrol remains incompletely understood, it is postulated that the compound acts as an inhibitor of specific enzymes, such as cyclooxygenase and lipoxygenase. Additionally, it is believed to possess antioxidant properties, potentially safeguarding cells against damage induced by free radicals.
Azo-Resveratrol (CAS 1393556-48-3) References
- Synthesis of novel azo-resveratrol, azo-oxyresveratrol and their derivatives as potent tyrosinase inhibitors. | Song, YM., et al. 2012. Bioorg Med Chem Lett. 22: 7451-5. PMID: 23142612
- A novel synthesized tyrosinase inhibitor: (E)-2-((2,4-dihydroxyphenyl)diazenyl)phenyl 4-methylbenzenesulfonate as an azo-resveratrol analog. | Bae, SJ., et al. 2013. Biosci Biotechnol Biochem. 77: 65-72. PMID: 23291747
- Evaluation of crocin and curcumin affinity on mushroom tyrosinase using surface plasmon resonance. | Patil, S., et al. 2014. Int J Biol Macromol. 65: 163-6. PMID: 24444880
- Design and discovery of mushroom tyrosinase inhibitors and their therapeutic applications. | Mendes, E., et al. 2014. Expert Opin Drug Discov. 9: 533-54. PMID: 24708040
- Natural, semisynthetic and synthetic tyrosinase inhibitors. | Lee, SY., et al. 2016. J Enzyme Inhib Med Chem. 31: 1-13. PMID: 25683082
- Skin whitening agents: medicinal chemistry perspective of tyrosinase inhibitors. | Pillaiyar, T., et al. 2017. J Enzyme Inhib Med Chem. 32: 403-425. PMID: 28097901
- Reversible optical control of F1 Fo -ATP synthase using photoswitchable inhibitors. | Eisel, B., et al. 2018. FEBS Lett. 592: 343-355. PMID: 29292505
- A comprehensive review on tyrosinase inhibitors. | Zolghadri, S., et al. 2019. J Enzyme Inhib Med Chem. 34: 279-309. PMID: 30734608
- Establishment of resveratrol and its derivatives as neuroprotectant against monocrotophos-induced alteration in NIPBL and POU4F1 protein through molecular docking studies. | Yadav, R. and Srivastava, P. 2020. Environ Sci Pollut Res Int. 27: 291-304. PMID: 31786755
- Design, synthesis, and biological evaluation of symmetrical azine derivatives as novel tyrosinase inhibitors. | Karimian, S., et al. 2021. BMC Chem. 15: 54. PMID: 34587988
- An updated organic classification of tyrosinase inhibitors on melanin biosynthesis. | Chen, C. Y., Lin, L. C., Yang, W. F., Bordon, J., & D Wang, H. M. (2015). Current Organic Chemistry,. 19(1):, 4-18.
- Synthetic routes of azo derivatives: a brief overview. | Shah, H. U. R., Ahmad, K., Naseem, H. A., Parveen, S., Ashfaq, M., Aziz, T.,.. & Shahzad, A. (2021. Journal of Molecular Structure,. 1244,: 131181.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Azo-Resveratrol, 10 mg | sc-503240 | 10 mg | $359.00 |
