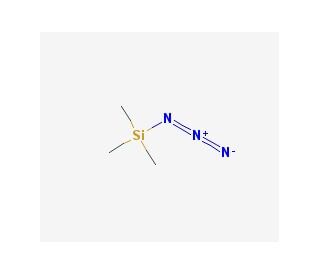
Molecular structure of Azidotrimethylsilane, CAS Number: 4648-54-8
Azidotrimethylsilane (CAS 4648-54-8)
Alternate Names:
Trimethylsilyl azide
Application:
Azidotrimethylsilane is a versatile azidonation reagent
CAS Number:
4648-54-8
Purity:
95%
Molecular Weight:
115.21
Molecular Formula:
C3H9N3Si
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Azidotrimethylsilane is a colorless, flammable liquid widely utilized as a multi-purpose reagent in organic chemistry. It reacts with nucleophiles to create azides, which can serve as precursors for additional compounds. Furthermore, Azidotrimethylsilane is involved in transfer reactions, including the formation of esters from trifluoroacetic acid and fatty acids. Azidotrimethylsilane is a preferred substitute. It is used in facilitating Schmidt rearrangement reactions and also serves as a nucleophile in the synthesis of organic compounds. Azidotrimethylsilane is a versatile azidonation reagent for amines, amides, aldehydes, and ketones.
Azidotrimethylsilane (CAS 4648-54-8) References
- A one-pot synthesis of tetrazolones from acid chlorides: understanding functional group compatibility, and application to the late-stage functionalization of marketed drugs. | Duncton, MA. and Singh, R. 2016. Org Biomol Chem. 14: 9338-9342. PMID: 27714179
- Synthesis of naphthyridin-2(1H)-one derivatives via ring expansion of 3-substituted-1H-pyrrolo[2,3-b]pyridin-2(3H)-one derivatives. | Croix, C., et al. 2018. Chem Commun (Camb). 54: 5538-5541. PMID: 29736506
- Copper-Catalyzed Synthesis of β-Azido Sulfonates or Fluorinated Alkanes: Divergent Reactivity of Sodium Sulfinates. | Xiong, Y., et al. 2018. Org Lett. 20: 6250-6254. PMID: 30246540
- Copper(I)-catalyzed tandem reaction: synthesis of 1,4-disubstituted 1,2,3-triazoles from alkyl diacyl peroxides, azidotrimethylsilane, and alkynes. | Israr, M., et al. 2018. Beilstein J Org Chem. 14: 2916-2922. PMID: 30546475
- MCR Scaffolds Get Hotter with 18F-Labeling. | Zarganes-Tzitzikas, T., et al. 2019. Molecules. 24: PMID: 30987302
- The Introduction of the Radical Cascade Reaction into Polymer Chemistry: A One-Step Strategy for Synchronized Polymerization and Modification. | Zhu, N., et al. 2020. iScience. 23: 100902. PMID: 32106054
- Visible-Light-Induced Photocatalyst-Free Aerobic Hydroxyazidations of Indoles: A Highly Regioselective and Stereoselective Synthesis of trans-2-Azidoindolin-3-ols. | Qiao, L., et al. 2021. J Org Chem. 86: 7955-7962. PMID: 34061526
- Asymmetric Azidation under Hydrogen Bonding Phase-Transfer Catalysis: A Combined Experimental and Computational Study. | Wang, J., et al. 2022. J Am Chem Soc. 144: 4572-4584. PMID: 35230845
- Biocompatible ionic liquid [Betaine][H2PO4] as a reusable catalyst for the substitution of xanthen-9-ol under solvent-free conditions. | Zhu, A., et al. 2020. RSC Adv. 10: 31662-31669. PMID: 35520689
- Electrochemical vicinal oxyazidation of α-arylvinyl acetates. | Li, YL., et al. 2022. Beilstein J Org Chem. 18: 1026-1031. PMID: 36051561
- Photoredox-catalyzed C-C bond cleavage of cyclopropanes for the formation of C(sp3)-heteroatom bonds. | Ge, L., et al. 2022. Nat Commun. 13: 5938. PMID: 36209214
- An electrochemical multicomponent reaction toward C-H tetrazolation of alkyl arenes and vicinal azidotetrazolation of alkenes. | Yu, Y., et al. 2022. Chem Sci. 13: 13851-13856. PMID: 36544744
- Development of Novel Pt(IV)-Carbohydrate Derivatives as Targeted Anticancer Agents against Osteosarcoma. | Moynihan, E., et al. 2023. Int J Mol Sci. 24: PMID: 37047001
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Azidotrimethylsilane, 10 g | sc-252397 | 10 g | $103.00 |
