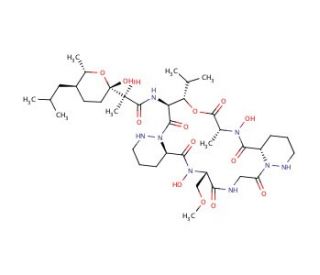
Aurantimycin A (CAS 162478-50-4)
QUICK LINKS
Aurantimycin A, identified with the CAS number 162478-50-4, is a member of the aurantimycin family of compounds, which are complex cyclic peptides produced by certain strains of marine bacteria. This compound features a unique macrocyclic structure incorporating multiple amino acids, which confer distinct biochemical properties and functionalities. The primary mechanism of action of Aurantimycin A revolves around its potent inhibitory effect on bacterial protein synthesis. It achieves this by binding to the bacterial ribosome, specifically interacting with the peptidyl transferase center of the 50S ribosomal subunit. This interaction disrupts the elongation process of protein synthesis, leading to an effective halt in bacterial growth and proliferation. In research, Aurantimycin A has been utilized as a tool to study bacterial ribosomal function and protein synthesis pathways. Its ability to selectively inhibit these processes in bacteria makes it a valuable compound for investigating the molecular mechanisms underlying protein synthesis and its regulation. Furthermore, studies involving Aurantimycin A contribute to the broader field of biochemistry by providing insights into the potential roles of cyclic peptides in natural biological control systems and their interactions with microbial communities. Through these research applications, Aurantimycin A enhances our understanding of microbial ecology and the biochemical strategies employed by marine bacteria.
Aurantimycin A (CAS 162478-50-4) References
- Pharmacological actions of a new synthetic cyclodepsipeptide, the A83586C-citropeptin hybrid, on complement C5a and G-Proteins. | Assem, ES., et al. 2008. Inflamm Res. 57 Suppl 1: S21-2. PMID: 18345507
- Mollemycin A: an antimalarial and antibacterial glyco-hexadepsipeptide-polyketide from an Australian marine-derived Streptomyces sp. (CMB-M0244). | Raju, R., et al. 2014. Org Lett. 16: 1716-9. PMID: 24611932
- Characterization of the aurantimycin biosynthetic gene cluster and enhancing its production by manipulating two pathway-specific activators in Streptomyces aurantiacus JA 4570. | Zhao, H., et al. 2016. Microb Cell Fact. 15: 160. PMID: 27655321
- Aurantimycin resistance genes contribute to survival of Listeria monocytogenes during life in the environment. | Hauf, S., et al. 2019. Mol Microbiol. 111: 1009-1024. PMID: 30648305
- PadR-type repressors controlling production of a non-canonical FtsW/RodA homologue and other trans-membrane proteins. | Hauf, S., et al. 2019. Sci Rep. 9: 10023. PMID: 31296881
- Elements in the LftR Repressor Operator Interface Contributing to Regulation of Aurantimycin Resistance in Listeria monocytogenes. | Hauf, S., et al. 2021. J Bacteriol. 203: PMID: 33649145
- Genome-Mining-Guided Discovery and Characterization of the PKS-NRPS-Hybrid Polyoxyperuin Produced by a Marine-Derived Streptomycete. | Kresna, IDM., et al. 2022. J Nat Prod. 85: 888-898. PMID: 35239335
- Potentiality of Actinomycetia Prevalent in Selected Forest Ecosystems in Assam, India to Combat Multi-Drug-Resistant Microbial Pathogens. | Mazumdar, R., et al. 2023. Metabolites. 13: PMID: 37623855
- Aurantimycins, new depsipeptide antibiotics from Streptomyces aurantiacus IMET 43917. Production, isolation, structure elucidation, and biological activity. | Gräfe, U., et al. 1995. J Antibiot (Tokyo). 48: 119-25. PMID: 7706121
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Aurantimycin A, 1 mg | sc-391347 | 1 mg | $60.00 |
