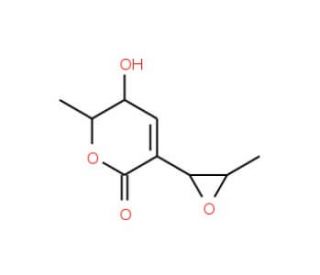
Aspyrone (CAS 17398-00-4)
QUICK LINKS
Aspyrone, bearing the CAS number 17398-00-4, is an organic compound categorized as a polyketide, which is produced by various species of the fungus Aspergillus. This compound is particularly interesting due to its structure, comprising a benzopyranone ring system, which is notable for its potential involvement in antioxidant activities within biological systems. The mechanism of action of aspyrone is primarily associated with its ability to act as an oxidative stress modulator. It is thought to interact with reactive oxygen species (ROS), potentially reducing oxidative damage in cellular components. This interaction makes aspyrone a valuable compound in research focused on understanding oxidative stress and its effects on cellular health and aging. Furthermore, aspyrone has been used in research to study inter-species chemical communication, particularly in fungal communities where such compounds may regulate or influence the behavior and development of other fungi. By examining the ecological role of aspyrone, researchers can gain insights into the natural roles of fungal secondary metabolites and their potential utility in managing fungal growth or behavior in various environments. Through these diverse applications, aspyrone contributes to a broader understanding of fungal ecology, secondary metabolite biosynthesis, and the potential protective mechanisms against oxidative stress.
Aspyrone (CAS 17398-00-4) References
- Fumiquinones A and B, nematicidal quinones produced by Aspergillus fumigatus. | Hayashi, A., et al. 2007. Biosci Biotechnol Biochem. 71: 1697-702. PMID: 17617730
- Chlorohydroaspyrones A and B, antibacterial aspyrone derivatives from the marine-derived fungus Exophiala sp. | Zhang, D., et al. 2008. J Nat Prod. 71: 1458-60. PMID: 18661951
- Cloning and characterization of novel methylsalicylic acid synthase gene involved in the biosynthesis of isoasperlactone and asperlactone in Aspergillus westerdijkiae. | Bacha, N., et al. 2009. Fungal Genet Biol. 46: 742-9. PMID: 19589392
- Development of an analytical method for identification of Aspergillus flavus based on chemical markers using HPLC-MS. | Saldan, NC., et al. 2018. Food Chem. 241: 113-121. PMID: 28958507
- Circumdatin-Aspyrone Conjugates from the Coral-Associated Aspergillus ochraceus LCJ11-102. | Fan, Y., et al. 2019. Mar Drugs. 17: PMID: 31284571
- Research Progress in the Biosynthetic Mechanisms of Marine Polyether Toxins. | Wan, X., et al. 2019. Mar Drugs. 17: PMID: 31652489
- Secondary Metabolites and Bioactivities of Aspergillus ochraceopetaliformis Isolated from Anthurium brownii. | Hu, HC., et al. 2020. ACS Omega. 5: 20991-20999. PMID: 32875235
- Production of 3-(1,2-epoxypropyl)-5,6-dihydro-5-hydroxy-6-methylpyran-2-one by Aspergillus ochraceus Wilhelm. | Moore, JH., et al. 1974. J Agric Food Chem. 22: 697-8. PMID: 4840919
- Synthesis of chiral aspyrone, a multi-functional dihydropyranone antibiotic. | Sugiyama, T., et al. 1995. Biosci Biotechnol Biochem. 59: 1921-4. PMID: 8534985
- Use of the maximum entropy method to correct for acoustic ringing and pulse breakthrough in 17O NMR spectra | Laue, E. D., et al. 1987. Journal of Magnetic Resonance. 72(3): 493-501.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Aspyrone, 1 mg | sc-202472 | 1 mg | $120.00 | |||
Aspyrone, 5 mg | sc-202472A | 5 mg | $450.00 |
