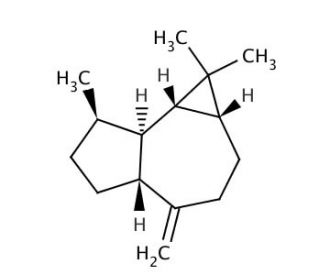
(+)-Aromadendrene (CAS 489-39-4)
QUICK LINKS
(+)-Aromadendrene, a sesquiterpene, is primarily of interest in the field of organic chemistry due to its complex molecular structure and unique chemical properties. This compound serves as a valuable subject in the study of sesquiterpene biosynthesis, offering insights into the enzymatic pathways and genetic regulation involved in the production of terpenoids. Research involving (+)-Aromadendrene can lead to a deeper understanding of the structural diversity and functional roles of terpenes in nature. Additionally, its involvement in synthetic chemistry research is noteworthy, as scientists explore methods to replicate and modify its structure for the development of new compounds with potential applications in various industries, including materials science and agriculture. The study of (+)-Aromadendrene also contributes to advancements in chemical ecology, where its interactions within ecosystems can provide valuable information on plant communication and defense mechanisms.
(+)-Aromadendrene (CAS 489-39-4) References
- First preparation of (+)-spathulenol. Regio- and stereoselective oxidation of (+)-aromadendrene with ozone. | FP Van Lier, TGM Hesp, LM Van Der Linde. 1985. Tetrahedron Letters. 26: 2109-2110.
- The conversion of natural (+)-aromadendrene into chiral synthons-I | HJM Gijsen, K Kanai, GA Stork, JBPA Wijnberg. 1990. Tetrahedron. 46: 7237-7246.
- Biotransformation of sesquiterpenoids, (+)-aromadendrene and (−)-alloaromadendrene by Glomerella cingulata | M Miyazawa, T Uemura, H Kameoka. 1995. Phytochemistry. 40: 793-796.
- Catalytic transformations of (+)-aromadendrene: Functionalization and isomerization reactions in the presence of the heteropoly acid catalyst H3PW12O40 | MSP Ribeiro, C de Souza Santos, CG Vieira. 2020. Molecular Catalysis. 498: 111264.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
(+)-Aromadendrene, 5 mg | sc-252389 | 5 mg | $152.00 |
