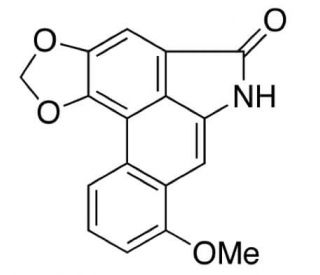
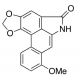
Aristolactam I (CAS 13395-02-3)
QUICK LINKS
Aristolactam I is a chemical compound that functions as an inhibitor of DNA topoisomerase II. It exerts its mechanism of action by binding to the enzyme and preventing the resealing of DNA strands that have been cleaved by the enzyme during its normal cellular processes. This interference with the enzyme′s activity leads to the accumulation of DNA strand breaks and ultimately inhibits cell proliferation. Aristolactam I may target the catalytic activity of DNA topoisomerase II, disrupting its essential role in DNA replication and transcription. Aristolactam I′s mechanism of action involves the inhibition of an important cellular process, making it useful for studying the role of DNA topoisomerases in cellular function.
Aristolactam I (CAS 13395-02-3) References
- Inhibition of renal NQO1 activity by dicoumarol suppresses nitroreduction of aristolochic acid I and attenuates its nephrotoxicity. | Chen, M., et al. 2011. Toxicol Sci. 122: 288-96. PMID: 21613233
- Aristolochic acid I metabolism in the isolated perfused rat kidney. | Priestap, HA., et al. 2012. Chem Res Toxicol. 25: 130-9. PMID: 22118289
- Isolation, structural elucidation and anti-inflammatory activity of astragalin, (-)hinokinin, aristolactam I and aristolochic acids (I & II) from Aristolochia indica. | Desai, DC., et al. 2014. Nat Prod Res. 28: 1413-7. PMID: 24854204
- Expression of Renal Aquaporins in Aristolochic Acid I and Aristolactam I-Induced Nephrotoxicity. | Li, J., et al. 2016. Nephron. 133: 213-21. PMID: 27352112
- Effect of base sequence context on the conformational heterogeneity of aristolactam-I adducted DNA: structural and energetic insights into sequence-dependent repair and mutagenicity. | Kathuria, P., et al. 2016. Toxicol Res (Camb). 5: 197-209. PMID: 30090337
- Quantitation of Protein Adducts of Aristolochic Acid I by Liquid Chromatography-Tandem Mass Spectrometry: A Novel Method for Biomonitoring Aristolochic Acid Exposure. | Chan, CK., et al. 2021. Chem Res Toxicol. 34: 144-153. PMID: 33410325
- Mitochondrial Iron Overload-Mediated Inhibition of Nrf2-HO-1/GPX4 Assisted ALI-Induced Nephrotoxicity. | Deng, HF., et al. 2020. Front Pharmacol. 11: 624529. PMID: 33584308
- An investigation on nephrotoxicity of Aristolactam I induced epithelial-mesenchymal transition on HK-2 cells. | Zhang, X., et al. 2021. Toxicon. 201: 21-26. PMID: 34391786
- Involvement of REV-ERBα dysregulation and ferroptosis in aristolochic acid I-induced renal injury. | Wang, Y., et al. 2021. Biochem Pharmacol. 193: 114807. PMID: 34673015
- Study of the Contents of Analogues of Aristolochic Acid in Houttuynia cordata by Ultra-High Performance Liquid Chromatography Tandem Mass Spectrometry. | Yu, X., et al. 2022. Foods. 11: PMID: 35159454
- Oral subacute nephrotoxicity of aristololactam I in rats. | Wang, C., et al. 2022. Toxicology. 475: 153228. PMID: 35690179
- Studies on the metabolism of aristolochic acids I and II. | Krumbiegel, G., et al. 1987. Xenobiotica. 17: 981-91. PMID: 3673113
- Bioaccumulation and DNA Adduct Formation of Aristolactam I: Unmasking a Toxicological Mechanism in the Pathophysiology of Aristolochic Acid Nephropathy. | Au, CK., et al. 2023. Chem Res Toxicol. 36: 322-329. PMID: 36757010
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Aristolactam I, 5 mg | sc-480598 | 5 mg | $430.00 |
