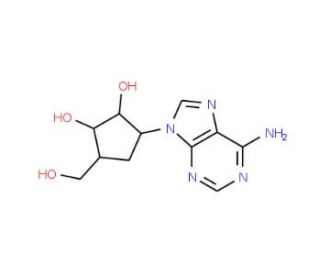
Molecular structure of Aristeromycin, CAS Number: 19186-33-5
Aristeromycin (CAS 19186-33-5)
See product citations (1)
Application:
Aristeromycin is an antibacterial adenosine analog
CAS Number:
19186-33-5
Molecular Weight:
265.27
Molecular Formula:
C11H15N5O3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Aristeromycin is a carbocyclic analogue of adenosine that exhibits antibacterial properties. Aristeromycin is derived from the nucleoside adenosine by replacing the ribose sugar with a cyclopent. Aristeromycin is an antibiotic and a potent S-adenosylhomocysteine hydrolase (AHCY) inhibitor. Aristeromycin exhibits antiviral and anticancer properties.
Aristeromycin (CAS 19186-33-5) References
- Highly stereoselective synthesis of aristeromycin through dihydroxylation of 4-aryl-1-azido-2-cyclopentenes. | Ainai, T., et al. 2004. J Org Chem. 69: 655-9. PMID: 14750788
- Synthesis and anti-HBV activity of α-stereoisomer of aristeromycin based analogs. | Kasula, M., et al. 2016. Bioorg Med Chem Lett. 26: 3945-9. PMID: 27426303
- Five-Membered Cyclitol Phosphate Formation by a myo-Inositol Phosphate Synthase Orthologue in the Biosynthesis of the Carbocyclic Nucleoside Antibiotic Aristeromycin. | Kudo, F., et al. 2016. Chembiochem. 17: 2143-2148. PMID: 27577857
- Aristeromycin and DZNeP cause growth inhibition of prostate cancer via induction of mir-26a. | Uchiyama, N., et al. 2017. Eur J Pharmacol. 812: 138-146. PMID: 28705714
- Coordinated Biosynthesis of the Purine Nucleoside Antibiotics Aristeromycin and Coformycin in Actinomycetes. | Xu, G., et al. 2018. Appl Environ Microbiol. 84: PMID: 30217843
- Nitrogen-Based Heterocyclic Compounds: A Promising Class of Antiviral Agents against Chikungunya Virus. | Santana, AC., et al. 2020. Life (Basel). 11: PMID: 33396631
- Synthesis and anti-HBV activity of carbocyclic nucleoside hybrids with salient features of entecavir and aristeromycin. | Samunuri, R., et al. 2020. RSC Med Chem. 11: 597-601. PMID: 33479662
- Differences in the metabolism and metabolic effects of the carbocyclic adenosine analogs, neplanocin A and aristeromycin. | Bennett, LL., et al. 1986. Mol Pharmacol. 29: 383-90. PMID: 3702857
- Interaction of ribonuclease from Aspergillus saitoi with aristeromycin and its phosphate esters. | Irie, M. 1971. J Biochem. 69: 965-8. PMID: 5577154
- Streptomyces citricolor nov. sp. and a new antibiotic, aristeromycin. | Kusaka, T., et al. 1968. J Antibiot (Tokyo). 21: 255-63. PMID: 5671989
- Induction of erythroid differentiation in leukaemic K562 cells by an S-adenosylhomocysteine hydrolase inhibitor, aristeromycin. | Mizutani, Y., et al. 1995. Biochem Biophys Res Commun. 207: 69-74. PMID: 7857307
- Effects of 4'-modified analogs of aristeromycin on the metabolism of S-adenosyl-L-homocysteine in murine L929 cells. | Ault-Riché, DB., et al. 1993. Mol Pharmacol. 43: 989-97. PMID: 8316227
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Aristeromycin, 5 mg | sc-233890 | 5 mg | $379.00 |
