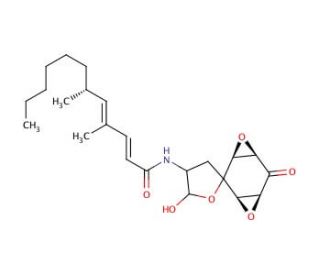
Aranorosin (CAS 117184-53-9)
QUICK LINKS
Aranorosin is a complex polyketide-derived natural product synthesized by the marine bacterium Streptomyces sp.. It belongs to the group of angucyclines, known for their distinctive angular tetracyclic ring structure, which plays a crucial role in their biological activity. Aranorosin exhibits a unique ability to interact with DNA, primarily through intercalation, a mechanism that disrupts the normal function of DNA by inserting itself between the base pairs of the DNA helix. This interaction inhibits DNA synthesis and transcription, making aranorosin a valuable tool for studying these fundamental biological processes. In research, aranorosin is employed to explore the dynamics of DNA interaction with small molecules, offering insights into the structural and functional implications of DNA binding. Its use extends to the study of transcriptional regulation, where it aids in understanding how transcription can be modulated by exogenous compounds. Moreover, aranorosin serves as a model compound for synthesizing new molecules with potential to regulate genetic expression, assisting in the development of novel approaches to manipulate these processes in various organisms. By investigating the properties of aranorosin, researchers are able to glean information on the broader class of angucyclines and their potential applications in biochemical and genetic engineering research.
Aranorosin (CAS 117184-53-9) References
- Aranorosinol A and aranorosinol B, two new metabolites from Pseudoarachniotus roseus: production, isolation, structure elucidation and biological properties. | Roy, K., et al. 1992. J Antibiot (Tokyo). 45: 1592-8. PMID: 1473986
- Aranorosin and a novel derivative inhibit the anti-apoptotic functions regulated by Bcl-2. | Nakashima, T., et al. 2008. Biochem Biophys Res Commun. 377: 1085-90. PMID: 18977202
- Aranorosin circumvents arbekacin-resistance in MRSA by inhibiting the bifunctional enzyme AAC(6')/APH(2″). | Suga, T., et al. 2012. J Antibiot (Tokyo). 65: 527-9. PMID: 22760297
- Potent Inhibitors of Acetyltransferase Eis Overcome Kanamycin Resistance in Mycobacterium tuberculosis. | Willby, MJ., et al. 2016. ACS Chem Biol. 11: 1639-46. PMID: 27010218
- Molecules that Inhibit Bacterial Resistance Enzymes. | Liu, Y., et al. 2018. Molecules. 24: PMID: 30583527
- Aranorosin, a novel antibiotic from Pseudoarachniotus roseus. I. Taxonomy, fermentation, isolation, chemical and biological properties. | Roy, K., et al. 1988. J Antibiot (Tokyo). 41: 1780-4. PMID: 3209472
- Aranorosin, a novel antibiotic from Pseudoarachniotus roseus. II. Structure elucidation. | Fehlhaber, HW., et al. 1988. J Antibiot (Tokyo). 41: 1785-94. PMID: 3209473
- Diastereo- and Enantioselective Cross-Couplings of Secondary Alkylcopper Reagents with 3-Halogeno-Unsaturated Carbonyl Derivatives. | Kremsmair, A., et al. 2020. Chemistry. 26: 11971-11973. PMID: 32557902
- Chemical investigations into the biosynthesis of the gymnastatin and dankastatin alkaloids. | Tong, B., et al. 2021. Chem Sci. 12: 8884-8891. PMID: 34257889
- Prospects for Antibacterial Discovery and Development. | Privalsky, TM., et al. 2021. J Am Chem Soc. 143: 21127-21142. PMID: 34860516
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Aranorosin, 250 µg | sc-391076 | 250 µg | $62.00 | |||
Aranorosin, 1 mg | sc-391076A | 1 mg | $188.00 |
