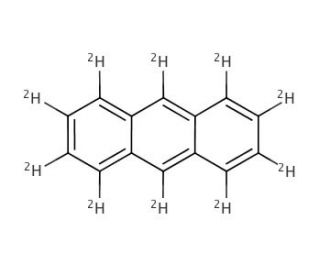
Anthracene-d10 (CAS 1719-06-8)
QUICK LINKS
Anthracene-d10, a deuterated compound belonging to the anthracene family, is an isotopically labeled form of anthracene. Through the replacement of hydrogen atoms with deuterium, a stable isotope of hydrogen, anthracene-d10 contains ten deuterium atoms within its solid crystalline structure, with a molecular formula of C14D10. With its unique isotopic composition, anthracene-d10 holds immense significance in scientific research, particularly in studies involving isotopic labeling and spectroscopy. Its incorporation of deuterium atoms allows anthracene-d10 to function as an internal reference or control in nuclear magnetic resonance (NMR) spectroscopy experiments, enabling precise measurements and facilitating comparisons. Consequently, it finds extensive applications in investigations involving NMR spectroscopy and other analytical techniques. Furthermore, anthracene-d10 plays a role in the development and calibration of analytical instruments like mass spectrometers and chromatographs. Its stable isotopic nature ensures accurate and reliable measurements, establishing it as a vital tool in chemical analysis and research. Beyond research applications, anthracene-d10 serves practical purposes across various industries. It is extensively employed in synthesizing labeled compounds for pharmaceutical research and development, wherein isotopic labeling plays a pivotal role in studying drug metabolism, pharmacokinetics, and tracing compound fate within biological systems. Moreover, anthracene-d10 has been utilized as a fluorescent probe for detecting DNA and RNA, a substrate for enzymes, and a fluorescent marker for cell imaging. Its interactions through hydrogen bonding and hydrophobic interactions enable it to bind with DNA, RNA, and proteins, facilitating its use as a versatile tool in various research areas such as studying protein structure and function, enzyme kinetics, and drug metabolism. In summary, anthracene-d10 serves as a valuable chemical tool, allowing for precise and reliable measurements in spectroscopic analysis, supporting advancements in scientific research, and facilitating the development of new pharmaceuticals. Its wide-ranging applications contribute significantly to diverse fields of study and industry needs.
Anthracene-d,[object Object], (CAS 1719-06-8) References
- Kinetics of OH radical reaction with anthracene and anthracene-d10. | Ananthula, R., et al. 2006. J Phys Chem A. 110: 3559-66. PMID: 16526636
- Validation of an Analytical Method for Determination of 13 priority polycyclic aromatic hydrocarbons in mineral water using dispersive liquid-liquid microextraction and GC-MS. | Sadeghi, R., et al. 2016. Iran J Pharm Res. 15: 157-68. PMID: 27610156
- Simultaneous Determination of 13 Priority Polycyclic Aromatic Hydrocarbons in Tehran's Tap Water and Water for Injection Samples Using Dispersive Liquid-Liquid Micro Extraction Method and Gas Chromatography-Mass Spectrometry. | Sadeghi, R., et al. 2016. Iran J Pharm Res. 15: 475-81. PMID: 27642318
- A Novel Liquid-Liquid Extraction for the Determination of Sertraline in Tap Water and Waste Water at Trace Levels by GC-MS. | Koçoğlu, ES., et al. 2017. Bull Environ Contam Toxicol. 99: 354-359. PMID: 28555336
- The gas-phase infrared spectra of anthracene-h10 and anthracene-d10 | , et al. (1997). J. Chem. Phys. 106,: 9004–9012.
- The raman spectrum of anthracene - d10 | A Bree, RA Kydd - Chemical Physics Letters, 1969 - Elsevier. June 1969,. Chemical Physics Letters. Volume 3, Issue 6,: Pages 357-360.
- Anharmonic Phonon Frequency Shifts and Widths in Solid Deuterated Anthracene-d10 | and J. F. J. Jordan, J. Kalus, U. Schmelzer, G. Eckold. September 1989. physica status solidi (b). Volume155, Issue1 1: Pages 89-105.
- Calculation and analysis of the excited states and fine structure of vibronic spectra of anthracene, anthracene-d 10, and tetracene | , et al. 2004). Optics and Spectroscopy. volume 96,: 335–342.
- Phosphorescence and triplet-triplet absorption spectra of anthracene-d10 in polymethylmethacrylate under high pressures☆ | RW Shaw, M Nicol - Chemical Physics Letters, 1976 - Elsevier. 1 April 1976,. Chemical Physics Letters. Volume 39, Issue 1,: Pages 108-112.
- Resonant radiofrequency magnetic field effects on a chemical reaction | JR Woodward, RJ Jackson, CR Timmel, PJ Hore… - Chemical physics …, 1997 - Elsevier. 4 July 1997. Chemical Physics Letters Volume. 272, Issues 5–6,,: Pages 376-382.
- Shift from an inverse to a normal isotope effect on the non-radiative decay rate of anthracene-d10 observed under partially jet-cooled conditions | H Niikura, S Hirayama - Chemical physics letters, 1998 - Elsevier. 6 November 1998,. Chemical Physics Letters. Volume 296, Issues 3–4,: Pages 343-349.
- Eigenvectors of low frequency internal phonons in crystalline anthracene-d10 | SL Chaplot, GS Pawley, EL Bokhenkov, EF Sheka… - Chemical Physics, 1981 - Elsevier. 15 May 1981. Chemical Physics,. Volume 57, Issue 3,: Pages 407-414.
- Correlation field splitting in anthracene crystals. The infra-red spectra of mixed crystals of anthracene and anthracene-D10 | S. Califano. 18 Sep 2006. Molecular Physics. Volume 5: 18 Sep 2006.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Anthracene-d10, 1 g | sc-252378 | 1 g | $130.00 | |||
Anthracene-d10, 5 g | sc-252378A | 5 g | $629.00 | |||
Anthracene-d10, 10 g | sc-252378B | 10 g | $1254.00 | |||
Anthracene-d10, 100 g | sc-252378C | 100 g | $12490.00 |
