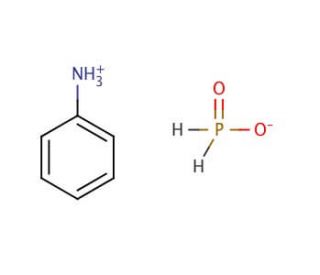
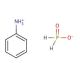
Molecular structure of Anilinium hypophosphite, CAS Number: 82395-88-8
Anilinium hypophosphite (CAS 82395-88-8)
Application:
Anilinium hypophosphite is a phosphorous containing compound
CAS Number:
82395-88-8
Purity:
97%
Molecular Weight:
159.12
Molecular Formula:
C6H10NO2P
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Anilinium hypophosphite functions as a reducing agent in various chemical reactions. Its mechanism of action involves donating electrons to other chemical species, leading to the reduction of those species and the oxidation of the hypophosphite ion. Anilinium Hypophosphite is utilized in the synthesis of metal nanoparticles, where it serves as a source of phosphorus and a reducing agent to facilitate the formation of nanoparticles with controlled size and morphology. Its ability to donate electrons and participate in redox reactions may be a useful component in the development of nanomaterials with tailored properties for various applications in materials science and catalysis.
Anilinium hypophosphite (CAS 82395-88-8) References
- Role of the nicotinic acid group in NAADP receptor selectivity. | Billington, RA., et al. 2005. Cell Calcium. 37: 81-6. PMID: 15541466
- Synthesis of p-aminophenyl aryl H-phosphinic acids and esters via cross-coupling reactions: elaboration to phosphinic acid pseudopeptide analogues of pteroyl glutamic acid and related antifolates. | Yang, Y. and Coward, JK. 2007. J Org Chem. 72: 5748-58. PMID: 17602593
- Palladium-catalyzed reactions of hypophosphorous compounds with allenes, dienes, and allylic electrophiles: methodology for the synthesis of allylic h-phosphinates. | Bravo-Altamirano, K., et al. 2008. J Org Chem. 73: 2292-301. PMID: 18275217
- Nickel-catalyzed C-P cross-coupling of diphenylphosphine oxide with aryl chlorides. | Zhang, HY., et al. 2012. Org Biomol Chem. 10: 9627-33. PMID: 23135327
- Recent Developments in the Addition of Phosphinylidene-Containing Compounds to Unactivated Unsaturated Hydrocarbons: Phosphorus-Carbon Bond-Formation via Hydrophosphinylation and Related Processes. | Coudray, L. and Montchamp, JL. 2008. European J Org Chem. 2008: 3601-3613. PMID: 23308039
- Synthesis of Monosubstituted Phosphinic Acids: Palladium-Catalyzed Cross-Coupling Reactions of Anilinium Hypophosphite | Jean-Luc Montchamp and Yves R. Dumond. 2001. J. Am. Chem. Soc., 123, 3,: 510–511.
- Direct synthesis of H-aryl and H-heteroarylphosphinic esters via palladium-catalyzed cross-coupling of alkylphosphinates | Z Huang, K Bravo-Altamirano, JL Montchamp - Comptes Rendus Chimie, 2004 - Elsevier. August–September 2004,. Comptes Rendus Chimie. Volume 7, Issues 8–9,: Pages 763-768.
- Development and Scale-Up of a Manufacturing Route for the Non-nucleoside Reverse Transcriptase Inhibitor GSK2248761A (IDX-899): Synthesis of an Advanced Key Chiral Intermediate | Richard Bellingham†§, Gary Borrett†, Guillaume Bret†§, Bernadette Choudary†§, David Colclough†§, Jerome Hayes†Orcid, John Hayler†Orcid, Neil Hodnett†, Alan Ironmonger*†, Augustine Ochen†, David Pascoe†, John Richardson†§, Erica Vit†§, François-René Alexandre‡Orcid, Catherine Caillet‡∥, Agnès Amador‡∥, Stéphanie Bot‡, Séverine Bonaric‡∥, Daniel da Costa‡, Marie-Pierre Lioure‡, Arlène Roland‡∥, Elodie Rosinovsky‡∥, Christophe Parsy‡∥, and Cyril B. Dousson‡. 2018,. Org. Process Res. Dev. 22, 2,: 200–206.
- Improving the efficiency and stability of tin-based perovskite solar cells using anilinium hypophosphite additive | Jorim Okoth Obila, ORCID logo a Hongwei Lei, ORCID logo *bc Elijah Omolo Ayieta,a Alex Awuor Ogacho,a Bernard O. Adudaa and Feng Wang ORCID logo b. 2021. New J. Chem.,.,45,: 8092-8100.
- Palladium-catalyzed cross-coupling reaction of anilinium hypophosphite with alkenyl bromides and triflates: application to the synthesis of GABA analogs | YR Dumond, JL Montchamp - Journal of organometallic chemistry, 2002 - Elsevier. 1 July 2002,. Journal of Organometallic Chemistry. Volume 653, Issues 1–2,: Pages 252-260.
- Enantioselective synthesis of phosphinyl peptidomimetics via an asymmetric Michael reaction of phosphinic acids with acrylate derivatives | X Liu, XE Hu, X Tian, A Mazur, FH Ebetino - Journal of organometallic …, 2002 - Elsevier. 1 March 2002,. Journal of Organometallic Chemistry. Volume 646, Issues 1–2,: Pages 212-222.
- Optoelectronic property refinement of FASnI3 films for photovoltaic application | JO Obila, H Lei, EO Ayieta, AA Ogacho, BO Aduda… - Materials Letters, 2021 - Elsevier. 1 October 2021,. Materials Letters. Volume 300,: 130099.
- Palladium-catalyzed phosphorus–carbon bond formation: cross-coupling reactions of alkyl phosphinates with aryl, heteroaryl, alkenyl, benzylic, and allylic halides and triflates | K Bravo-Altamirano, Z Huang, JL Montchamp - Tetrahedron, 2005 - Elsevier. 27 June 2005,. Tetrahedron. Volume 61, Issue 26,: Pages 6315-6329.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Anilinium hypophosphite, 25 g | sc-233875 | 25 g | $72.00 |
