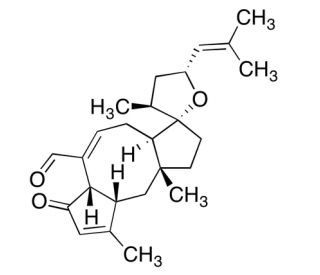
Anhydroophiobolin A (CAS 6026-65-9)
QUICK LINKS
Anhydroophiobolin A is a sesterterpene compound noted for its involvement in the inhibition of calmodulin-dependent cyclic nucleotide phosphodiesterase, highlighting its utility in cellular signaling studies. The chemical operates by targeting the calmodulin pathway, a critical signaling route in various organisms, which modulates diverse cellular functions through calcium signaling. This mechanism is pivotal in research focused on understanding intracellular communication and the regulation of calcium-dependent pathways. Anhydroophiobolin A has been utilized extensively in biochemistry research to dissect the roles of calmodulin in cellular processes, including calcium signaling, protein phosphorylation, and gene expression, providing insights into the fundamental mechanisms governing cell physiology. Additionally, its role in inhibiting phosphodiesterase makes it a valuable tool in studying the dynamics of cyclic nucleotide concentrations within cells, further contributing to our understanding of cellular response mechanisms to various stimuli. This chemical has thus been a crucial agent in experimental setups aimed at unraveling the complexities of cellular signaling pathways and their implications in cellular function and behavior.
Anhydroophiobolin A (CAS 6026-65-9) References
- Isolation of ophiobolin A and its analogs as inhibitors to photosynthesis[J]. | Kim J M, Hyeon S B, Isogai A,., 1984,. Agricultural and biological chemistry. 48(3):: 803-805.
- Phytotoxins produced by germinating spores of Bipolaris oryzae[J]. | **ao J Z, Tsuda M, Doke N,. 1991. Phytopathology,., 81(1):: 58-64.
- The taxonomy and biology of dematiaceous hyphomycetes and their mycotoxins[J]. | Sivanesan A. 1991. Fungi and mycotoxins in stored products,. (36):: 47-64.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Anhydroophiobolin A, 1 mg | sc-391809 | 1 mg | $325.00 |
