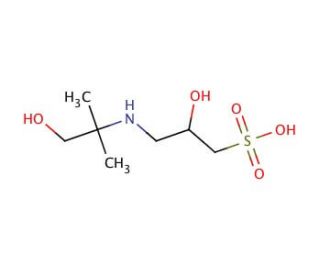
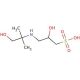
AMPSO (CAS 68399-79-1)
QUICK LINKS
AMPSO (2-Amino-2-methyl-1-propanol sulfonic acid) is a zwitterionic biological buffer that is extensively used in biochemical and molecular biology research to stabilize pH in biological systems, particularly under conditions where other common buffers might fail. AMPSO has a useful buffering range of 8.3 to 10.1, making it particularly suitable for alkaline applications. Its buffering capacity is crucial in maintaining a stable environment during biochemical reactions that are sensitive to pH fluctuations, such as DNA/RNA extractions and protein solubility studies at higher pH levels. The chemical structure of AMPSO, featuring both amino and sulfonic acid groups, allows it to operate efficiently as a zwitterionic buffer; it can donate or accept protons, thus stabilizing pH by mitigating changes caused by acidic or basic compounds in the solution. This unique property is essential for applications such as electrophoresis and chromatographic techniques where precise pH control is necessary to ensure accurate mobility and separation of biomolecules. In research settings, AMPSO is valued not only for its pH stability but also for its low UV absorbance and minimal interaction with biological molecules, ensuring that it does not interfere with the biochemical processes being studied. This makes AMPSO an indispensable tool in laboratories focusing on high-pH biochemistry and molecular biology protocols.
AMPSO (CAS 68399-79-1) References
- Quantum chemical studies on structural, vibrational, NBO and hyperpolarizability of N-(1,1-Dimethyl-2-hydroxyethyl)-3-amino-2-hydroxypropanesulfonic acid. | Renuga Devi, TS., et al. 2015. Spectrochim Acta A Mol Biomol Spectrosc. 136 Pt C: 1500-14. PMID: 25459712
- Kinetic Study of CO2 Hydration by Small-Molecule Catalysts with A Second Coordination Sphere that Mimic the Effect of the Thr-199 Residue of Carbonic Anhydrase. | Park, D. and Lee, MS. 2019. Biomimetics (Basel). 4: PMID: 31581538
- Kinetic study of catalytic CO2 hydration by metal-substituted biomimetic carbonic anhydrase model complexes. | Park, D. and Lee, MS. 2019. R Soc Open Sci. 6: 190407. PMID: 31598240
- The Impact of Redox, Hydrolysis and Dehydration Chemistry on the Structural and Magnetic Properties of Magnetoferritin Prepared in Variable Thermal Conditions. | Balejčíková, L., et al. 2021. Molecules. 26: PMID: 34834056
- Metabolomic Profiling Reveals New Insight of Fowl Adenovirus Serotype 4 Infection. | Ma, H. and Niu, Y. 2021. Front Microbiol. 12: 784745. PMID: 35111140
- Allosteric coupling between transmembrane segment 4 and the selectivity filter of TALK1 potassium channels regulates their gating by extracellular pH. | Tsai, WH., et al. 2022. J Biol Chem. 298: 101998. PMID: 35500647
- Acidic Shift of Optimum pH of Bovine Serum Amine Oxidase upon Immobilization onto Nanostructured Ferric Tannates. | Rilievo, G., et al. 2022. Int J Mol Sci. 23: PMID: 36293026
- Anion-Exchange Chromatography at the Service of Gene Therapy: Baseline Separation of Full/Empty Adeno-Associated Virus Capsids by Screening of Conditions and Step Gradient Elution Mode. | Aebischer, MK., et al. 2022. Int J Mol Sci. 23: PMID: 36293189
- Role of Human Aldo-Keto Reductases in the Nitroreduction of 1-Nitropyrene and 1,8-Dinitropyrene. | Su, AL., et al. 2022. Chem Res Toxicol. 35: 2296-2309. PMID: 36399404
- Crystal Structure, Steady-State, and Pre-Steady-State Kinetics of Acinetobacter baumannii ATP Phosphoribosyltransferase. | Read, BJ., et al. 2024. Biochemistry. 63: 230-240. PMID: 38150593
- Thermodynamic quantities for the ionization reactions of buffers | Goldberg, R. N., Kishore, N., & Lennen, R. M. 2002. Journal of physical and chemical reference data. 31(2): 231-370.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
AMPSO, 25 g | sc-239247 | 25 g | $65.00 |
