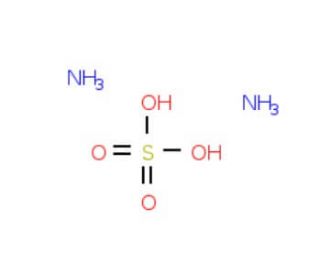
Ammonium sulfate solution, 3.2 M (CAS 7783-20-2)
QUICK LINKS
Ammonium sulfate solution, with a concentration of 3.2 molar (3.2M), is a valuable chemical reagent utilized across various scientific disciplines, including biochemistry, molecular biology, and protein purification. This solution is prepared by dissolving ammonium sulfate salt in water to achieve the desired concentration, resulting in a clear or slightly cloudy liquid. In scientific research, ammonium sulfate solution serves multiple purposes owing to its diverse properties and functionalities. One of its primary applications is in protein purification and crystallization. Ammonium sulfate is a widely used salt for protein precipitation due to its ability to selectively precipitate proteins based on their solubility at different salt concentrations. By adjusting the concentration of ammonium sulfate in solution, researchers can effectively separate and purify proteins from complex mixtures. Moreover, ammonium sulfate solution finds applications in nucleic acid purification, particularly in DNA and RNA extraction protocols. It is often used to precipitate nucleic acids from aqueous solutions, facilitating their isolation and purification from cellular debris and contaminants. Additionally, ammonium sulfate solution is employed as a stabilizing agent and cryoprotectant in various biochemical and cell culture applications. It helps maintain the stability and activity of enzymes, proteins, and other biomolecules during storage and experimental procedures. Furthermore, ammonium sulfate solution is utilized in molecular biology techniques such as chromatography, electrophoresis, and DNA sequencing. Its compatibility with these techniques makes it a versatile reagent in laboratory protocols for the separation, analysis, and manipulation of biomolecules.
Ammonium sulfate solution, 3.2 M (CAS 7783-20-2) References
- A preliminary X-ray crystallographic study of spinach cytochrome C. | Morita, Y., et al. 1973. FEBS Lett. 31: 186-188. PMID: 11947110
- High performance liquid chromatographic determination of acetoacetate by post-column derivatization with p-nitrobenzene diazonium fluoroborate. | Yamato, S., et al. 2003. Biol Pharm Bull. 26: 397-400. PMID: 12673014
- Isolation, purification and characterization of hemerythrin from Methylococcus capsulatus (Bath). | Kao, WC., et al. 2008. J Inorg Biochem. 102: 1607-14. PMID: 18397812
- Purification, crystallization and preliminary X-ray diffraction analysis of the seryl-tRNA synthetase from Candida albicans. | Rocha, R., et al. 2011. Acta Crystallogr Sect F Struct Biol Cryst Commun. 67: 153-6. PMID: 21206050
- Using shaped ultrafast laser pulses to detect enzyme binding. | Tseng, CH., et al. 2011. Opt Express. 19: 24638-46. PMID: 22109492
- Complex of myoglobin with phenol bound in a proximal cavity. | Huang, X., et al. 2012. Acta Crystallogr Sect F Struct Biol Cryst Commun. 68: 1465-71. PMID: 23192025
- Oligosaccharide-based Surfactant/Citric Acid Buffer System Stabilizes Lactate Dehydrogenase during Freeze-drying and Storage without the Addition of Natural Sugar. | Ogawa, S., et al. 2016. J Oleo Sci. 65: 525-32. PMID: 27181251
- Stereochemical course of DNA hydrolysis by nuclease S1. | Potter, BV., et al. 1983. J Biol Chem. 258: 1758-60. PMID: 6296110
- Contribution to the study in solution and solid state of the rabbit aldolase temperature dependence. | Jollès, P., et al. 1980. FEBS Lett. 116: 48-50. PMID: 7409136
- Structural studies on the loggerhead sea turtle (Caretta caretta) myoglobin. | Petruzzelli, R., et al. 1993. Biochem Mol Biol Int. 31: 19-24. PMID: 8260943
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ammonium sulfate solution, 3.2 M, 100 ml | sc-291897 | 100 ml | $16.00 | |||
Ammonium sulfate solution, 3.2 M, 500 ml | sc-291897A | 500 ml | $36.00 |
