Ammonium oxalate monohydrate (CAS 6009-70-7)
QUICK LINKS
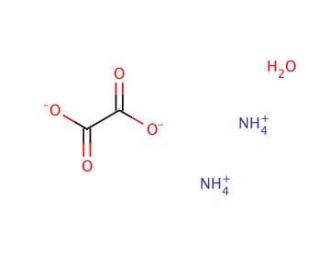
Ammonium oxalate monohydrate, with the chemical formula (NH4)2C2O4·H2O, is a white crystalline salt that is highly soluble in water. This compound is the ammonium salt of oxalic acid, and it is frequently utilized in analytical chemistry for its ability to effectively precipitate metal ions. The primary mechanism of action of ammonium oxalate monohydrate involves its reaction with metal cations to form insoluble oxalates, which are crucial in the qualitative and quantitative analysis of metal ions in solutions. This precipitation reaction is particularly useful in the determination of calcium ions in biochemistry as well as in environmental and geological samples, where accurate measurement of metal content is essential.
Ammonium oxalate monohydrate (CAS 6009-70-7) References
- Immunochemical detection of oxalate monoalkylamide, an ascorbate-derived Maillard reaction product in the human lens. | Nagaraj, RH., et al. 1999. FEBS Lett. 453: 327-30. PMID: 10405169
- Evaluation of an acid ammonium oxalate extraction to determine fluoride resident concentrations in soils. | Bégin, L. and Fortin, J. 2003. J Environ Qual. 32: 662-73. PMID: 12708691
- Comparison of the ammonium oxalate extraction of fertilizer for K2O, method 983.02, with the ammonium citrate/EDTA extraction, method 993.31. | Newlon, NF. 2003. J AOAC Int. 86: 640-2. PMID: 14509419
- [Determination of cadmium in soil slurry samples by GFAAS using ammonium oxalate as disperstant]. | Gan, W., et al. 1999. Guang Pu Xue Yu Guang Pu Fen Xi. 19: 861-3. PMID: 15822318
- Negative Linear Compressibility in Organic Mineral Ammonium Oxalate Monohydrate with Hydrogen Bonding Wine-Rack Motifs. | Qiao, Y., et al. 2015. J Phys Chem Lett. 6: 2755-60. PMID: 26266859
- Platelet surface interaction in suspensions of rabbit blood in ammonium oxalate. | Grüner, OP. 1970. Scand J Haematol. 7: 274-8. PMID: 5477541
- Experimental acute ammonium oxalate poisoning of sheep. | James, MP., et al. 1971. Aust Vet J. 47: 9-17. PMID: 5542376
- On the Effect of Cu(II) Impurity on the Growth Kinetics of Ammonium Oxalate Monohydrate Crystals from Aqueous Solutions | K. Sangwal Prof. dr hab., E. Mielniczek-Brzóska Dr. 2001. Crystal Research and Technology. 36: 837-849.
- Effect of impurities on metastable zone width for the growth of ammonium oxalate monohydrate crystals from aqueous solutions | K. Sangwal a, E. Mielniczek-Brzóska b. 2004. Journal of Crystal Growth. 267: 662-675.
- Growth kinetics of ammonium oxalate monohydrate single crystals from aqueous solutions containing Co(II) and Ni(II) impurities | E. Mielniczek-Brzóska, K. Sangwal. 2004. Crystal Research and Technology. 39: 993-1005.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ammonium oxalate monohydrate, 500 g | sc-202945 | 500 g | $64.00 | |||
Ammonium oxalate monohydrate, 2.5 kg | sc-202945A | 2.5 kg | $231.00 | |||
Ammonium oxalate monohydrate, 100 g | sc-202945B | 100 g | $42.00 |
