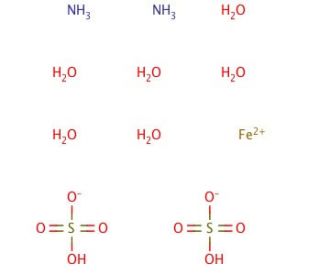

Ammonium iron(II) sulfate hexahydrate (CAS 7783-85-9)
QUICK LINKS
Ammonium iron(II) sulfate hexahydrate or Mohr′s Salt is often employed as an analytical standard, and has been used in a variety of other applications from nanomaterials to general redox reactions. This product is much less affected by oxygen in the air than iron(II) sulfate (sc-228358), making it more desirable for titration purposes, where iron(II) might be oxidized to iron(III). The oxidation of iron(II) to iron(III) is pH dependent, and the reaction occurs more rapidly at higher pH Mohr′s Salt lowers the pH of solutions slightly, thereby preventing oxidation from occurring. The significance of ammonium iron(II) sulfate hexahydrate extends to scientific research, serving as a vital reagent in diverse biochemical and physiological experiments. It plays a pivotal role in studying the impact of iron on biological systems, conducting iron detection in biological samples, and catalyzing organic synthesis reactions. Additionally, it finds application in the analysis of proteins, lipids, carbohydrates, and other biomolecules. By acting as a proton donor, it influences the pH of its surroundings, thereby affecting the activity of enzymes and other proteins. Furthermore, it acts as a Lewis acid, accepting electrons from other molecules and thereby influencing the structure and reactivity of the involved molecules. Ammonium iron(II) sulfate hexahydrate, or ferrous sulfate, represents a versatile compound with a wide range of applications in scientific exploration, industrial processes, and medical contexts. Its ability to modulate pH and interact with various molecules allows for valuable insights and advancements in numerous fields of study.
Ammonium iron(II) sulfate hexahydrate (CAS 7783-85-9) References
- Bivoltametric titrations using electrodes with innovative geometry. | Surmann, P., et al. 1996. Anal Bioanal Chem. 356: 192-6. PMID: 15048351
- Ammonia formation by the reduction of nitrite/nitrate by FeS: ammonia formation under acidic conditions. | Summers, DP. 2005. Orig Life Evol Biosph. 35: 299-312. PMID: 16228644
- Catalytic effect of different forms of iron in purification of single-walled carbon nanotubes. | Suzuki, T., et al. 2010. J Nanosci Nanotechnol. 10: 3924-8. PMID: 20355392
- Online monitoring applying the anaerobic respiratory monitoring system reveals iron(II) limitation in YTF medium for Clostridium ljungdahlii. | Mann, M., et al. 2021. Eng Life Sci. 21: 19-28. PMID: 33531887
- Magnetic Separation and Centri-Chronoamperometric Detection of Foodborne Bacteria Using Antibiotic-Coated Metallic Nanoparticles. | Diouani, MF., et al. 2021. Biosensors (Basel). 11: PMID: 34201531
- Whole Three-Dimensional Dosimetry of Carbon Ion Beams with an MRI-Based Nanocomposite Fricke Gel Dosimeter Using Rapid T1 Mapping Method. | Mizukami, S., et al. 2021. Gels. 7: PMID: 34940293
- Comparative Studies to Uncover Mechanisms of Action of N-(1,3,4-Oxadiazol-2-yl)benzamide Containing Antibacterial Agents. | Naclerio, GA., et al. 2022. ACS Infect Dis. 8: 865-877. PMID: 35297603
- Production and characterization of chitooligosaccharides by the fungal chitinase Chit42 immobilized on magnetic nanoparticles and chitosan beads: selectivity, specificity and improved operational utility. | Kidibule, PE., et al. 2021. RSC Adv. 11: 5529-5536. PMID: 35423100
- Copper-Induced Interactions of Caffeic Acid and Sinapic Acid to Generate New Compounds in Artificial Biological Fluid Conditions. | Iwasaki, Y., et al. 2022. Antioxidants (Basel). 11: PMID: 35883798
- Regulation of Iron-Ion Transporter SLC11A2 by Three Identical miRNAs. | Sugino, Y., et al. 2022. Biol Pharm Bull. 45: 1291-1299. PMID: 36047197
- Evaluation of Malondialdehyde Levels, Oxidative Stress and Host-Bacteria Interactions: Escherichia coli and Salmonella Derby. | Tsaturyan, V., et al. 2022. Cells. 11: PMID: 36230950
- Carbamoyltransferase Enzyme Assay: In vitro Modification of 5-hydroxymethylcytosine (5hmC) to 5-carbamoyloxymethylcytosine (5cmC). | Yang, W., et al. 2022. Bio Protoc. 12: PMID: 36245800
- Phenylalanine hydroxylase mRNA rescues the phenylketonuria phenotype in mice. | Cacicedo, ML., et al. 2022. Front Bioeng Biotechnol. 10: 993298. PMID: 36277393
- Development of a New Colorimetric, Kinetic and Automated Ceruloplasmin Ferroxidase Activity Measurement Method. | Neşelioğlu, S., et al. 2022. Antioxidants (Basel). 11: PMID: 36358559
- Effect of Al and Fe Doping on the Electrochemical Behavior of Li1.2Ni0.133Mn0.534Co0.133O2 Li-Rich Cathode Material. | Medvedeva, A., et al. 2022. Materials (Basel). 15: PMID: 36431711
- Adsorptive removal of tetracycline and ciprofloxacin drugs from water by using a magnetic rod-like hydroxyapatite and MIL-101(Fe) metal-organic framework nanocomposite. | Beiranvand, M., et al. 2022. RSC Adv. 12: 34438-34453. PMID: 36545621
- Visible-Light-Enhanced Antibacterial Activity of Silver and Copper Co-Doped Titania Formed on Titanium via Chemical and Thermal Treatments. | Suzuki, K., et al. 2023. Molecules. 28: PMID: 36677708
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ammonium iron(II) sulfate hexahydrate, 100 g | sc-202940 | 100 g | $61.00 | |||
Ammonium iron(II) sulfate hexahydrate, 500 g | sc-202940A | 500 g | $122.00 |
