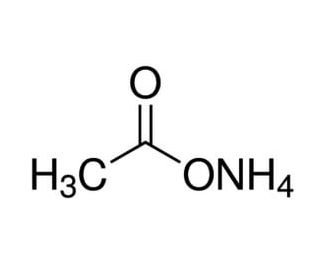
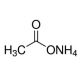
Ammonium acetate solution, 5M (CAS 631-61-8)
QUICK LINKS
Ammonium acetate solution, a concentrated solution containing ammonium acetate dissolved in water, is a versatile chemical reagent with widespread applications in scientific research. This solution is meticulously prepared to provide a high concentration of ammonium acetate, typically 5 molar (5M), for use in various experimental procedures. In research, ammonium acetate solution serves as a buffering agent, pH regulator, and source of ammonium ions. Its buffering capacity and ability to maintain a stable pH make it valuable in biochemical and molecular biology experiments, particularly in nucleic acid isolation, purification, and precipitation. Ammonium acetate is often used in conjunction with ethanol for the precipitation of DNA and RNA from aqueous solutions, facilitating the removal of contaminants and purification of nucleic acids. Moreover, ammonium acetate solution finds applications in protein crystallization, mass spectrometry, and chromatography techniques such as ion exchange chromatography and reverse-phase chromatography. Its ability to selectively precipitate proteins and peptides, as well as its compatibility with various analytical methods, make it a versatile reagent in protein biochemistry and structural biology research. Furthermore, ammonium acetate solution is employed in chemical synthesis reactions as a source of acetate ions and a mild base catalyst. It is utilized in organic synthesis, including esterification, acetylation, and amidation reactions, where acetate ions participate as nucleophiles or leaving groups.
Ammonium acetate solution, 5M (CAS 631-61-8) References
- [Pneumococcal Vulvovaginitis and Its Relationship to Pneumococcal Peritonitis]. | WINKLER, A. 1949. Klin Med Osterr Z Wiss Prakt Med. 4: 269-74. PMID: 18149302
- Loss of the Habenula Intrinsic Neuromodulator Kisspeptin1 Affects Learning in Larval Zebrafish. | Lupton, C., et al. 2017. eNeuro. 4: PMID: 28534042
- Radioimmunotherapy of methicillin-resistant Staphylococcus aureus in planktonic state and biofilms. | van Dijk, B., et al. 2020. PLoS One. 15: e0233086. PMID: 32407350
- The DNase I sensitive domain of the chicken lysozyme gene spans 24 kb. | Jantzen, K., et al. 1986. Nucleic Acids Res. 14: 6085-99. PMID: 3748804
- Formation of compact globular particles in interphase nuclei from rat liver under the effect of polyanions. | Prusov, AN., et al. 1993. Biochem Biophys Res Commun. 193: 591-8. PMID: 8512560
- Genomic yeast DNA clone banks. Construction and gene isolation. | Bignell, GR. and Evans, IH. 1996. Methods Mol Biol. 53: 155-87. PMID: 8924978
- Stereoselective synthesis of butenolide and butyrolactone derivatives. | SIRIT, ABDULKADİR. 2000. Turkish Journal of Chemistry. 24.2: 185-190.
- Evaluation of a Real time polymerase chain reaction assay for the detection of aflatoxin/sterigmatocystin producing strains of Aspergillus spp. | Singh, R., et al. 2017. Indian Journal of Animal Research. 51.4: 676-678.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ammonium acetate solution, 5M, 100 ml | sc-291892 | 100 ml | $118.00 | |||
Ammonium acetate solution, 5M, 250 ml | sc-291892A | 250 ml | $232.00 | |||
Ammonium acetate solution, 5M, 1 L | sc-291892B | 1 L | $374.00 |
