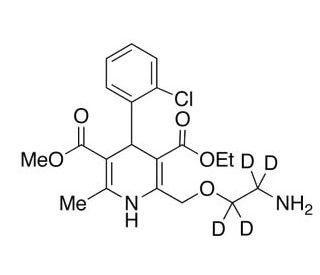
Amlodipine-d4 (CAS 1185246-14-3)
QUICK LINKS
Amlodipine-d4 is a deuterium-labeled version of amlodipine. The presence of deuterium atoms can increase the molecular stability of amlodipine-d4, making it less susceptible to metabolic degradation. This property is particularly useful in analytical chemistry, where amlodipine-d4 can serve as an internal standard in mass spectrometry (MS) or nuclear magnetic resonance (NMR) spectroscopy. These analytical techniques benefit from the use of deuterium-labeled compounds because the slight difference in mass between deuterium and hydrogen allows for precise tracking and quantification of the compound and its metabolites in biological samples. In kinetic research, amlodipine-d4 enables scientists to study the absorption, distribution, metabolism, and excretion (ADME) of amlodipine with high accuracy. By comparing the behavior of the labeled and unlabeled versions of amlodipine, researchers can gain insights into how the compound is processed.
Amlodipine-d4 (CAS 1185246-14-3) References
- Simple RP-HPLC method for determination of triple drug combination of valsartan, amlodipine and hydrochlorothiazide in human plasma. | Sharma, RN. and Pancholi, SS. 2012. Acta Pharm. 62: 45-58. PMID: 22472448
- [Simultaneous determination of amlodipine, benazepril and benazeprilat in human plasma by LC-HESI/MS/MS method]. | Pan, HL., et al. 2014. Yao Xue Xue Bao. 49: 95-100. PMID: 24783513
- Development and Validation of a LC-MS/MS Method for the Simultaneous Estimation of Amlodipine and Valsartan in Human Plasma: Application to a Bioequivalence Study. | Jangala, H., et al. 2014. Sci Pharm. 82: 585-600. PMID: 25853070
- Simultaneous determination of rosuvastatin and amlodipine in human plasma using tandem mass spectrometry: Application to disposition kinetics. | Narapusetti, A., et al. 2015. J Adv Res. 6: 931-40. PMID: 26644931
- Factors Influencing the Relationship Between the Dose of Amlodipine Required for Blood Pressure Control and Change in Blood Pressure in Hypertensive Cats. | Bijsmans, ES., et al. 2016. J Vet Intern Med. 30: 1630-1636. PMID: 27717190
- Evaluation of Pharmacokinetic Interactions Between Lesinurad, a New Selective Urate Reabsorption Inhibitor, and CYP Enzyme Substrates Sildenafil, Amlodipine, Tolbutamide, and Repaglinide. | Gillen, M., et al. 2017. Clin Pharmacol Drug Dev. 6: 363-376. PMID: 28067999
- Therapeutic drug monitoring of amlodipine and the Z-FHL/HHL ratio: Adherence tools in patients referred for apparent treatment-resistant hypertension. | Jones, ESW., et al. 2017. S Afr Med J. 107: 887-891. PMID: 29022534
- Pharmacokinetic comparison of a fixed-dose combination versus concomitant administration of fimasartan, amlodipine, and rosuvastatin using partial replicated design in healthy adult subjects. | Oh, M., et al. 2018. Drug Des Devel Ther. 12: 1157-1164. PMID: 29780236
- Quantification of 21 antihypertensive drugs in serum using UHPLC-MS/MS. | Gundersen, POM., et al. 2018. J Chromatogr B Analyt Technol Biomed Life Sci. 1089: 84-93. PMID: 29783197
- Pharmacokinetic and Safety Profiles of a Fixed-Dose Combination of Amlodipine, Valsartan, and Atorvastatin: A 3-Period Replicate Crossover Study. | Kim, S., et al. 2020. Clin Pharmacol Drug Dev. 9: 386-394. PMID: 31373174
- Postmortem fatal and non-fatal concentrations of amlodipine. | Alvarez, JC., et al. 2020. Forensic Sci Int. 316: 110555. PMID: 33152659
- Mechanistic Considerations About an Unexpected Ramipril Drug-Drug Interaction in the Development of a Triple Fixed-Dose Combination Product Containing Ramipril, Amlodipine, and Atorvastatin. | Hermann, R., et al. 2021. Clin Pharmacol Drug Dev. 10: 1307-1315. PMID: 33773093
- Pharmacokinetics and Bioequivalence Evaluation of 2 Olmesartan Medoxomil and Amlodipine Besylate Fixed-Dose Combination Tablets in Healthy Chinese Volunteers Under Fasting and Fed Conditions. | Li, X., et al. 2022. Clin Pharmacol Drug Dev. 11: 761-769. PMID: 35289500
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Amlodipine-d4, 5 mg | sc-481432 | 5 mg | $347.00 | |||
Amlodipine-d4, 50 mg | sc-481432A | 50 mg | $2448.00 |
