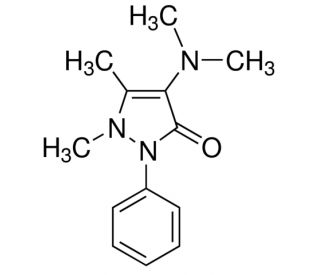
Molecular structure of Aminopyrine, CAS Number: 58-15-1
Aminopyrine (CAS 58-15-1)
Alternate Names:
Aminophenazone; 4-Dimethylaminoantipyrine
CAS Number:
58-15-1
Purity:
≥98%
Molecular Weight:
231.29
Molecular Formula:
C13H17N3O
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Aminopyrine is an organic compound classified as an aminophenol, featuring both an amine group and a phenol group connected by a single bond in its aromatic structure. Its applications in the synthesis of diverse organic compounds are widespread. Furthermore, it serves as a versatile reagent and catalyst in organic chemistry for the creation of various organic compounds. Additionally, it finds use as a chromogenic agent in detecting specific proteins. Research suggests that it may act as an inhibitor of the enzyme monoamine oxidase (MAO), responsible for the breakdown of essential neurotransmitters like serotonin and dopamine.
Aminopyrine (CAS 58-15-1) References
- Metabolism and excretion of aminopyrine in man. | Goromaru, T., et al. 1976. Chem Pharm Bull (Tokyo). 24: 1376-83. PMID: 1021292
- Aminopyrine: metabolism and effects in the rat after administration of inhibitors of hepatic monooxygenases. | Di Nucci, A., et al. 1979. Eur J Drug Metab Pharmacokinet. 4: 179-83. PMID: 118879
- AMINOPYRINE, DIPYRONE AND AGRANULOCYTOSIS. | KATZ, A. 1964. Can Med Assoc J. 91: 1229-30. PMID: 14226100
- Enzymatic dealkylation of aminopyrine (pyramidon) and other alkylamines. | LA DU, BN., et al. 1955. J Biol Chem. 214: 741-5. PMID: 14381412
- Further metabolism of 4-acetylaminoantipyrine, the major metabolite of aminopyrine, in rats. | Tanaka, T., et al. 1987. Chem Pharm Bull (Tokyo). 35: 3519-22. PMID: 3427730
- Studies on the biliary efflux of GSH from rat liver due to the metabolism of aminopyrine. | Krieter, PA., et al. 1985. Biochem Pharmacol. 34: 955-60. PMID: 3985999
- Urine data analysis for pharmacokinetics of aminopyrine and its metabolites in man. | Koizumi, T., et al. 1974. Chem Pharm Bull (Tokyo). 22: 894-906. PMID: 4419027
- Effects of aminopyrine and phenylbutazone on the activity of neurons in the trigeminal nuclei and hypothalamus. | Sasa, M., et al. 1969. Jpn J Pharmacol. 19: 363-72. PMID: 5307469
- Evaluation and comparison of colorimetric, radiometric and high performance liquid chromatographic assays for aminopyrine-N-demethylation by rat liver microsomes. | Bast, A. and Noordhoek, J. 1981. J Pharm Pharmacol. 33: 14-8. PMID: 6114145
- The measurement of plasma concentration of aminopyrine and its metabolites in man. | Goromaru, T., et al. 1978. Chem Pharm Bull (Tokyo). 26: 33-7. PMID: 639167
- Interpretation of CO2 exhalation rate data from demethylation of aminopyrine and its metabolite monomethylaminoantipyrine. | Rhodes, JC., et al. 1982. Br J Clin Pharmacol. 14: 409-14. PMID: 6812609
- Bacterial degradation of aminopyrine. | Blecher, H., et al. 1981. Xenobiotica. 11: 749-54. PMID: 7336755
- Metabolism of aminopyrine and derivatives in man: in vivo study of monomorphic and polymorphic metabolic pathways. | Agúndez, JA., et al. 1995. Xenobiotica. 25: 417-27. PMID: 7645307
- Oxidation of aminopyrine by hypochlorite to a reactive dication: possible implications for aminopyrine-induced agranulocytosis. | Uetrecht, JP., et al. 1995. Chem Res Toxicol. 8: 226-33. PMID: 7766805
- Determination of aminopyrine, dipyrone and its metabolites in urine by high-performance liquid chromatography. | Agúndez, JA., et al. 1994. Ther Drug Monit. 16: 316-22. PMID: 8085285
Activator of:
CPO.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Aminopyrine, 50 g | sc-484806 | 50 g | $67.00 | |||
Aminopyrine, 250 g | sc-484806A | 250 g | $333.00 |
