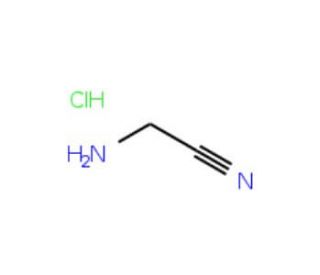
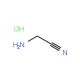
Aminoacetonitrile hydrochloride (CAS 6011-14-9)
QUICK LINKS
Aminoacetonitrile is a useful synthetic intermediate with diverse applications. It is a reagent for the synthesis of dipeptide nitriles, with reversible and potent inhibitory activity against cathepsin S. Additionally, aminoacetonitrile is used in the preparation of substituted cyclic ureas as potential inhibitors of HIV protease. Aminoacetonitrile hydrochloride acts as a strong base in organic synthesis reactions. Its strong basicity enables it to protonate weakly acidic compounds and catalyze the formation of carbon-nitrogen bonds. Furthermore, it plays a significant role in catalyzing the formation of amine-containing compounds, including amines, amides, and nitriles. it is capable of forming hydrogen bonds with other molecules, which facilitates the formation of polymers and various materials.
Aminoacetonitrile hydrochloride (CAS 6011-14-9) References
- Variation in aspects of cysteine proteinase catalytic mechanism deduced by spectroscopic observation of dithioester intermediates, kinetic analysis and molecular dynamics simulations. | Reid, JD., et al. 2001. Biochem J. 357: 343-52. PMID: 11439083
- Diastereoselective arylithium addition to an alpha-trifluoromethyl imine. Practical synthesis of a potent cathepsin K inhibitor. | Roy, A., et al. 2006. J Org Chem. 71: 4320-3. PMID: 16709080
- Cyclocondensation of alpha-aminonitriles and enones: a short access to 3,4-dihydro-2H-pyrrole 2-carbonitriles and 2,3,5-trisubstituted pyrroles. | Bergner, I., et al. 2009. J Org Chem. 74: 8243-53. PMID: 19785394
- Cyanomethanaminium tetra-fluoro-borate. | Han, MT. and Zhang, Y. 2010. Acta Crystallogr Sect E Struct Rep Online. 66: o1941. PMID: 21588267
- Cyano-methanaminium perchlorate. | Quan, J. 2012. Acta Crystallogr Sect E Struct Rep Online. 68: o3480. PMID: 23476288
- Molecular Design, Synthesis and Trypanocidal Activity of Dipeptidyl Nitriles as Cruzain Inhibitors. | Avelar, LA., et al. 2015. PLoS Negl Trop Dis. 9: e0003916. PMID: 26173110
- Discovery of a Flexible Triazolylbutanoic Acid as a Highly Potent Uric Acid Transporter 1 (URAT1) Inhibitor. | Tian, H., et al. 2016. Molecules. 21: PMID: 27854343
- Lithocholic Acid Hydroxyamide Destabilizes Cyclin D1 and Induces G0/G1 Arrest by Inhibiting Deubiquitinase USP2a. | Magiera, K., et al. 2017. Cell Chem Biol. 24: 458-470.e18. PMID: 28343940
- Preparation and biological properties of 2-thio-containing pyrimidines and their condensed analogs. | Voskoboynik, OY., et al. 2017. Chem Heterocycl Compd (N Y). 53: 256-272. PMID: 32214419
- Synthesis of Biologically Relevant 1,2,3- and 1,3,4-Triazoles: From Classical Pathway to Green Chemistry. | Gonnet, L., et al. 2021. Molecules. 26: PMID: 34577138
- Nitrification Progress of Nitrogen-Rich Heterocyclic Energetic Compounds: A Review. | Luo, Y., et al. 2022. Molecules. 27: PMID: 35268569
- Di-, tetra-, and perhydropyrrolo[1,2-a]imidazoles: The Methods of Synthesis and Some Aspects of Application. | Saliyeva, LM., et al. 2022. Chem Heterocycl Compd (N Y). 58: 661-680. PMID: 36687505
- The alpha and gamma subunits of initiation factor eIF-2 can be cross-linked to 18S ribosomal RNA within the quaternary initiation complex, eIF-2.Met-tRNAf.GDPCP.small ribosomal subunit. | Westermann, P., et al. 1980. Nucleic Acids Res. 8: 3065-71. PMID: 7443514
- Effect of a lathyrogen (aminoacetonitrile hydrochloride) on aortic physiosclerosis and skeletal mineralization in growing rats of different age. | Morgan, AJ. and Bellamy, D. 1976. Age Ageing. 5: 155-63. PMID: 973590
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Aminoacetonitrile hydrochloride, 25 g | sc-278693 | 25 g | $72.00 |
