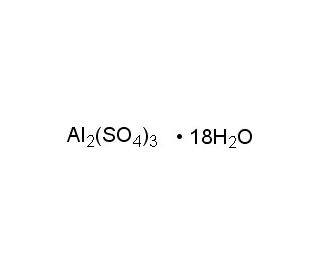
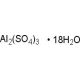
Aluminum sulfate octadecahydrate (CAS 7784-31-8)
QUICK LINKS
Aluminum sulfate octadecahydrate, a hydrous form of aluminum sulfate, is a white crystalline solid widely recognized for its utility in environmental engineering and industrial applications. This chemical serves as a pivotal coagulant in water treatment processes; its primary action involves the neutralization of the electrical charges on particles within the water. This neutralization causes the particles to aggregate and settle, consequently clarifying the water by removing turbidity and other impurities. Such a process is essential not only for making water more suitable for consumption but also for its use in various industrial processes. In environmental research, aluminum sulfate octadecahydrate is often studied for its efficacy in reducing phosphate levels in wastewater, thereby aiding in the control of eutrophication in bodies of water. This helps maintain ecological balance and prevents the excessive growth of algae. Additionally, in the paper manufacturing industry, it is employed in the paper sizing process to enhance the water resistance of paper. This compound′s role extends into academic and industrial research, where it contributes to the optimization of coagulation processes and paper production, as well as the exploration of its interactions with both organic and inorganic substances in various applications.
Aluminum sulfate octadecahydrate (CAS 7784-31-8) References
- Solubility controls on aluminum in drinking water at relatively low and high pH. | Kvech, S. and Edwards, M. 2002. Water Res. 36: 4356-68. PMID: 12420940
- Alterations in electrocardiographic parameters after subacute exposure of fluoride and ameliorative action of aluminium sulphate in goats. | Kant, V., et al. 2010. Biol Trace Elem Res. 134: 188-94. PMID: 19652927
- Aluminum polymers formed following alum treatment of lake water. | Anderson, MA. and Berkowitz, J. 2010. Chemosphere. 81: 832-6. PMID: 20825969
- Phase transformations of α-alumina made from waste aluminum via a precipitation technique. | Matori, KA., et al. 2012. Int J Mol Sci. 13: 16812-21. PMID: 23222685
- Solubility of Ammonium Aluminum Sulfate Dodecahydrate in the Aluminum Sulfate + Ammonium Sulfate + Sulfuric Acid + Water System at (25, 30, 40, and 50) °C | Věra Hostomská and Jiří Hostomský. 2006. J. Chem. Eng. Data. 51: 243–247.
- Ion association and hydration in 3:2 electrolyte solutions by dielectric spectroscopy: Aluminum sulfate | Simon Schrödle a 1, Wolfram W. Rudolph b, Glenn Hefter a, Richard Buchner c. 2007. Geochimica et Cosmochimica Acta. 71: 5287-5300.
- Evaluation of cellulose content in hemp shives after salt catalyzed hydrolysis | Prans Brazdausks a b, Maris Puke b, Janis Rizhikovs b, Jelena Pubule a. 2017. Energy Procedia. 128: 297-301.
- Study on kinetics of the pyrolysis process of aluminum sulfate | Guangya Zheng, Jupei Xia, Zhengjie Chen, Jin Yang & Chenglong Liu. 2020. Phosphorus, Sulfur, and Silicon and the Related Elements. 195: 285-292.
- A new approach of aluminum extraction from drinking water treatment sludge using ammonium sulfate roasting process | Ibtissam Ballou a, Said Kounbach b, Jamal Naja a, Zine Elabidine Bakher c, Khalid Laraki d, Fatima Raibi d, Radouan Saadi d, Sanae Kholtei a. 2022. Minerals Engineering. 189.
- Tuning interfacial relaxations in P(VDF-HFP) with Al2O3@ZrO2 core-shell nanofillers for enhanced dielectric and energy storage performance | Wenyue Zheng a 1, Lulu Ren a 1, Xuetong Zhao a, He Li b, Zongliang Xie c, Yupeng Li a, Can Wang a, Liang Yu a, Lijun Yang a, Ruijin Liao a. 2022. Composites Science and Technology. 222.
- New insight into the combined effect of aluminum sulfate and triethanolamine on cement hydration | Liheng Zhang a, Zichen Lu a, Yanrong Zhang b, Zhiwei Liu a, Ye Pan a, Guoqing Zhang c, Yan He d, Zhenping Sun a. 2024. Cement and Concrete Research. 181.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Aluminum sulfate octadecahydrate, 100 g | sc-202928 | 100 g | $48.00 | |||
Aluminum sulfate octadecahydrate, 500 g | sc-202928A | 500 g | $184.00 | |||
Aluminum sulfate octadecahydrate, 2.5 kg | sc-202928B | 2.5 kg | $673.00 |
