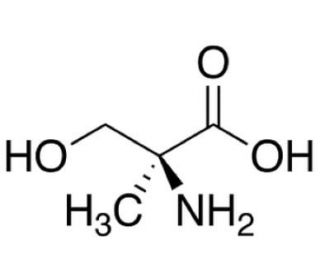
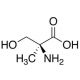
α-Methyl-L-serine (CAS 16820-18-1)
QUICK LINKS
α-Methyl-L-serine is used in biochemical to study enzyme kinetics and substrate specificity, particularly in reactions involving amino acid metabolism. In enzymology, α-Methyl-L-serine acts as a competitive inhibitor for various enzymes that normally process L-serine, thereby helping to active sites and mechanism of enzyme action. Å-Methyl-L-Serine is designed to understand the structural requirements for substrate binding and catalysis in enzymatic reactions.On α-Methyl-L-serine also extends to its use in probing the effects of structural changes on the biological functions of amino acids.Studies involving α-Methyl-L-serine contribute to the field of protein engineering, where it is used to investigate the impact of amino acid substitutions on protein stability and function, offering insights into protein design and function prediction.
α-Methyl-L-serine (CAS 16820-18-1) References
- Gene cloning of alpha-methylserine aldolase from Variovorax paradoxus and purification and characterization of the recombinant enzyme. | Nozaki, H., et al. 2008. Biosci Biotechnol Biochem. 72: 2580-8. PMID: 18838814
- Purification and gene cloning of alpha-methylserine aldolase from Ralstonia sp. strain AJ110405 and application of the enzyme in the synthesis of alpha-methyl-L-serine. | Nozaki, H., et al. 2008. Appl Environ Microbiol. 74: 7596-9. PMID: 18952881
- Biosynthesis of the 4-methyloxazoline-containing nonribosomal peptides, JBIR-34 and -35, in Streptomyces sp. Sp080513GE-23. | Muliandi, A., et al. 2014. Chem Biol. 21: 923-34. PMID: 25041948
- Structural and Functional Analyses of the Tridomain-Nonribosomal Peptide Synthetase FmoA3 for 4-Methyloxazoline Ring Formation. | Katsuyama, Y., et al. 2021. Angew Chem Int Ed Engl. 60: 14554-14562. PMID: 33783097
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
α-Methyl-L-serine, 50 mg | sc-219472 | 50 mg | $255.00 |
