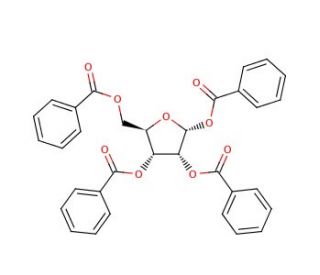
α-D-Ribofuranose 1,3,5-tribenzoate (CAS 22224-41-5)
QUICK LINKS
α-D-Ribofuranose 1,3,5-tribenzoate is a chemically modified form of ribofuranose, wherein the hydroxyl groups at the 1, 3, and 5 positions are esterified with benzoate groups. This modification significantly enhances the molecule′s stability and solubility in organic solvents, making it an invaluable tool in organic synthesis and nucleoside research. The tribenzoate groups protect the ribofuranose during synthetic manipulations, preventing unwanted side reactions and facilitating the introduction of other functional groups in a controlled manner. Researchers utilize this compound primarily to study the synthesis of modified nucleosides and nucleotides, which are fundamental components of RNA. These studies are crucial for understanding the chemical properties of ribonucleic acid components and exploring new pathways for synthesizing RNA analogs. The chemical′s ability to serve as a protected intermediate enables scientists to construct more complex molecules systematically, which is essential for the development of novel biochemical probes and materials. Additionally, α-D-Ribofuranose 1,3,5-tribenzoate is used in the preparation of labeled ribofuranoses for spectroscopic studies, helping to explain the structure and dynamics of carbohydrate molecules in various chemical environments, thus advancing fundamental research in carbohydrate chemistry and glycoscience.
α-D-Ribofuranose 1,3,5-tribenzoate (CAS 22224-41-5) References
- 2'-C-branched ribonucleosides. 2. Synthesis of 2'-C-beta-trifluoromethyl pyrimidine ribonucleosides. | Li, NS., et al. 2001. Org Lett. 3: 1025-8. PMID: 11277786
- 2'-C-Branched Ribonucleosides: Synthesis of the Phosphoramidite Derivatives of 2'-C-beta-Methylcytidine and Their Incorporation into Oligonucleotides. | Tang, XQ., et al. 1999. J Org Chem. 64: 747-754. PMID: 11674142
- Distribution of 1-(2-deoxy-2-fluoro-beta-D-arabinofuranosyl) uracil in mice bearing colorectal cancer xenografts: rationale for therapeutic use and as a positron emission tomography probe for thymidylate synthase. | Eiseman, JL., et al. 2004. Clin Cancer Res. 10: 6669-76. PMID: 15475457
- Synthesis of 2'-C-difluoromethylribonucleosides and their enzymatic incorporation into oligonucleotides. | Ye, JD., et al. 2005. J Org Chem. 70: 7902-10. PMID: 16277309
- Synthesis of 2'-C-beta-methyl-2'-deoxyguanosine. | Li, NS., et al. 2009. J Org Chem. 74: 2227-30. PMID: 19173609
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
α-D-Ribofuranose 1,3,5-tribenzoate, 5 g | sc-257035 | 5 g | $185.00 |
