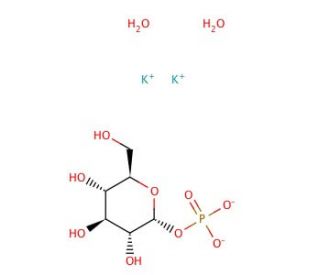
α-D-Glucose 1-phosphate dipotassium salt (CAS 5996-14-5)
QUICK LINKS
α-D-Glucose 1-phosphate dipotassium salt is a widely used chemical compound in scientific research. It is derived from glucose, a vital source of energy for living organisms. One of its primary uses is as a substrate for enzyme assays, particularly for studying enzymes involved in glycogen metabolism. Additionally, it plays a role in the synthesis of various important molecules. In scientific research, α-D-Glucose 1-phosphate dipotassium salt serves as a substrate for measuring enzyme activity, particularly those related to glycogen metabolism. Moreover, it is utilized in the synthesis of essential molecules like nucleotides and glycosaminoglycans. The mechanism of action of α-D-Glucose 1-phosphate dipotassium salt stems from its ability to act as a substrate for enzymes involved in glycogen metabolism. Through the action of the enzyme phosphoglucomutase, Alpha-Glucose-1-phosphate is converted into glucose-6-phosphate, which can further be metabolized to provide energy for cellular processes.
α-D-Glucose 1-phosphate dipotassium salt (CAS 5996-14-5) References
- Structural analysis of amylose tris(3,5-dimethylphenylcarbamate) by NMR relevant to its chiral recognition mechanism in HPLC. | Yamamoto, C., et al. 2002. J Am Chem Soc. 124: 12583-9. PMID: 12381203
- NMR and computational studies of chiral discrimination by amylose tris(3,5-dimethylphenylcarbamate). | Ye, YK., et al. 2007. J Phys Chem B. 111: 1189-98. PMID: 17266274
- Immobilized polysaccharide derivatives: chiral packing materials for efficient HPLC resolution. | Ikai, T., et al. 2007. Chem Rec. 7: 91-103. PMID: 17394175
- [Optimization of enzymatic preparation of glucose 1-phosphate by response surface methodology]. | Wang, X., et al. 2013. Sheng Wu Gong Cheng Xue Bao. 29: 107-10. PMID: 23631123
- Synthesis and application of immobilized polysaccharide-based chiral stationary phases for enantioseparation by high-performance liquid chromatography. | Shen, J., et al. 2014. J Chromatogr A. 1363: 51-61. PMID: 24997110
- Preparation of silica gel-bonded amylose through enzyme-catalyzed polymerization and chiral recognition ability of its phenylcarbamate derivative in HPLC. | Enomoto, N., et al. 1996. Anal Chem. 68: 2798-804. PMID: 8794916
- Enantioseparation using selected polysaccharides as chiral buffer additives in capillary electrophoresis. | Chankvetadze, B., et al. 1997. J Chromatogr A. 773: 331-8. PMID: 9228803
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
α-D-Glucose 1-phosphate dipotassium salt, 2 g | sc-222432 | 2 g | $198.00 | |||
α-D-Glucose 1-phosphate dipotassium salt, 5 g | sc-222432A | 5 g | $303.00 |
