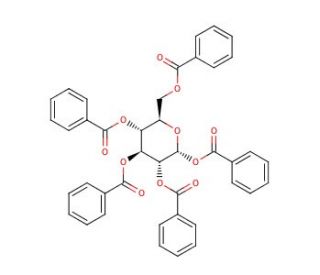
α-D-Glucopyranose pentabenzoate (CAS 22415-91-4)
QUICK LINKS
α-D-Glucopyranose pentabenzoate is a compound that functions as a protecting group in organic synthesis. It is used to temporarily mask the hydroxyl groups of glucose, preventing unwanted reactions at these sites during chemical transformations. The mechanism of action involves the formation of ester linkages between the benzoate groups and the hydroxyl groups of the glucose molecule. This modification alters the reactivity of the hydroxyl groups, allowing specific chemical reactions to occur without interference from other parts of the molecule. α-D-Glucopyranose pentabenzoate plays a role in controlling the regioselectivity and stereoselectivity of reactions involving glucose derivatives, making it useful in the synthesis of complex organic molecules. Its mechanism of action at the molecular level involves the reversible formation of covalent bonds with the hydroxyl groups, providing a means to manipulate α-D-Glucopyranose pentabenzoate reactivity of glucose for experimental purposes.
α-D-Glucopyranose pentabenzoate (CAS 22415-91-4) References
- Synthesis and structure-activity relationship study of antidiabetic penta-O-galloyl-D-glucopyranose and its analogues. | Ren, Y., et al. 2006. J Med Chem. 49: 2829-37. PMID: 16640344
- Stereoselective glycosylations using benzoylated glucosyl halides with inexpensive promoters. | Murakami, T., et al. 2008. Carbohydr Res. 343: 1297-308. PMID: 18406402
- Lack of activation of UCP1 in isolated brown adipose tissue mitochondria by glucose-O-ω-modified saturated fatty acids of various chain lengths. | Breen, EP., et al. 2013. J Chem Biol. 6: 121-33. PMID: 24432128
- Glycosylation of Nucleosides. | Zhang, Y. and Knapp, S. 2016. J Org Chem. 81: 2228-42. PMID: 26938666
- Stereoselective glycosylations using benzoylated glucosyl halides with inexpensive promoters. | Murakami, Teiichi, et al. 2008. Carbohydrate research. 343.8: 1297-1308.
- Simplified beta-glycosylation of peptides. | Zhang, Yonglian and Spencer Knapp. 2018. Tetrahedron. 74.23: 2891-2903.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
α-D-Glucopyranose pentabenzoate, 1 g | sc-257033 | 1 g | $21.00 |
