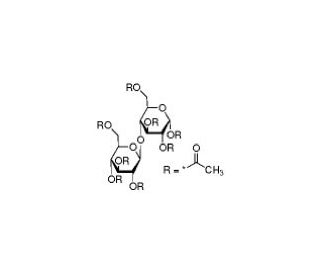
α-D-Cellobiose octaacetate (CAS 5346-90-7)
QUICK LINKS
α-D-Cellobiose octaacetate is a derivative of cellobiose, a disaccharide composed of two glucose units linked by a β(1→4) glycosidic bond, with all hydroxyl groups acetylated. This acetylation enhances its solubility in organic solvents, facilitating its use in various research applications. Primarily, α-D-Cellobiose octaacetate is employed in carbohydrate chemistry and enzymology studies. It serves as a substrate for glycosidase enzymes, allowing researchers to investigate the catalytic mechanisms and specificity of cellulases and other glycosidases. These studies are crucial for understanding cellulose degradation, which has significant implications for biofuel production and biomass utilization. Additionally, α-D-Cellobiose octaacetate is used in synthetic organic chemistry as a building block for the preparation of more complex oligosaccharides and glycoconjugates. Researchers utilize it to study the conformational properties of cellobiose derivatives, exploring their interactions with various chemical and biological systems. This compound also plays a role in studying the binding affinities and interactions of carbohydrate-binding proteins, such as lectins, which are involved in cell-cell recognition and signaling processes. Overall, α-D-Cellobiose octaacetate is a valuable tool in advancing our understanding of carbohydrate chemistry, enzyme mechanisms, and the structural biology of glycoconjugates.
α-D-Cellobiose octaacetate (CAS 5346-90-7) References
- 4-Methyl-7-thioumbelliferyl-beta-D-cellobioside: a fluorescent, nonhydrolyzable substrate analogue for cellulases. | Barr, BK. and Holewinski, RJ. 2002. Biochemistry. 41: 4447-52. PMID: 11914092
- Ruminiclostridium josui Abf62A-Axe6A: A tri-functional xylanolytic enzyme exhibiting α-l-arabinofuranosidase, endoxylanase, and acetylxylan esterase activities. | Wang, Y., et al. 2018. Enzyme Microb Technol. 117: 1-8. PMID: 30037546
- Copper-Catalyzed Anomeric O-Arylation of Carbohydrate Derivatives at Room Temperature. | Verdelet, T., et al. 2019. J Org Chem. 84: 9226-9238. PMID: 31274301
- Rapid cyclic ion mobility separations of monosaccharide building blocks as a first step toward a high-throughput reaction screening platform for carbohydrate syntheses. | Peterson, TL. and Nagy, G. 2021. RSC Adv. 11: 39742-39747. PMID: 35494126
- Self-Assembly of Unusually Stable Thermotropic Network Phases by Cellobiose-Based Guerbet Glycolipids. | Das, S., et al. 2024. Biomacromolecules. 25: 1291-1302. PMID: 38170593
- Commercial synthesis of alpha-D-cellobiosyl bromide heptaacetate. | Goggin, KD., et al. 1994. J Chem Technol Biotechnol. 60: 253-6. PMID: 7764992
- CPMAS 13C NMR and X‐ray studies of cellooligosaccharide acetates as a model for cellulose triacetate. | Kono, Hiroyuki, et al. 1999. Journal of Polymer Science Part A: Polymer Chemistry. 37.22: 4100-4107.
- Induction of cellulases by disaccharides or their derivatives in Penicillium janthinellum EMS-UV-8 mutant. | Agrawal, Ruchi, et al. 2017. Biofuels. 8.5: 615-622.
- Penicillium: the next emerging champion for cellulase production. | Vaishnav, Neha, et al. 2018. Bioresource Technology Reports. 2: 131-140.
- Characterization and quantitation of steryl glycosides in solanum melongena. | Heinz, Philipp and Marcus A. Glomb. 2018. Journal of agricultural and food chemistry. 66.43: 11398-11406.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
α-D-Cellobiose octaacetate, 25 g | sc-257032 | 25 g | $107.00 |
