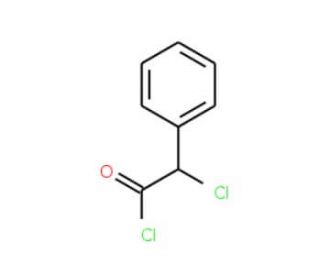
α-Chlorophenylacetyl chloride (CAS 2912-62-1)
QUICK LINKS
α-Chlorophenylacetyl chloride is a chemical compound that functions as an acylating agent in organic synthesis. It is used to introduce the α-chlorophenylacetyl group into various organic molecules, allowing for the modification of their chemical properties. The mechanism of action involves the acylation of nucleophilic functional groups, such as hydroxyl or amino groups, through the formation of covalent bonds. This acylation reaction results in the formation of new compounds with altered reactivity and properties, which can be further utilized in the development of novel materials intermediates. At the molecular level, α-Chlorophenylacetyl chloride interacts with specific functional groups within the target molecules, leading to the formation of acylated products. The mechanism of action enables the precise modification of organic compounds, contributing to the diversification of chemical libraries and the exploration of structure-activity relationships in development.
α-Chlorophenylacetyl chloride (CAS 2912-62-1) References
- A solid-phase approach to mouse melanocortin receptor agonists derived from a novel thioether cyclized peptidomimetic scaffold. | Bondebjerg, J., et al. 2002. J Am Chem Soc. 124: 11046-55. PMID: 12224952
- Farnesyltransferase inhibitors inhibit the growth of malaria parasites in vitro and in vivo. | Wiesner, J., et al. 2004. Angew Chem Int Ed Engl. 43: 251-4. PMID: 14695623
- Novel C6-substituted 1,3,4-oxadiazinones as potential anti-cancer agents. | Alam, MM., et al. 2015. Oncotarget. 6: 40598-610. PMID: 26515601
- Recent Developments in General Methodologies for the Synthesis of α-Ketoamides. | De Risi, C., et al. 2016. Chem Rev. 116: 3241-305. PMID: 26881454
- Discovery of Indole Derivatives as Novel and Potent Dengue Virus Inhibitors. | Bardiot, D., et al. 2018. J Med Chem. 61: 8390-8401. PMID: 30149709
- Recent Advances in the Synthesis of Complex Macromolecular Architectures Based on Poly(N-vinyl pyrrolidone) and the RAFT Polymerization Technique. | Roka, N., et al. 2022. Polymers (Basel). 14: PMID: 35215614
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
α-Chlorophenylacetyl chloride, 10 g | sc-239154 | 10 g | $292.00 |
