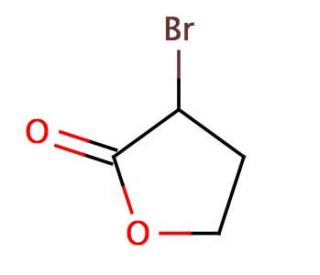
Molecular structure of α-Bromo-γ-butyrolactone, CAS Number: 5061-21-2
α-Bromo-γ-butyrolactone (CAS 5061-21-2)
Alternate Names:
2-Bromo-4-hydroxybutyric acid γ-lactone
Application:
α-Bromo-γ-butyrolactone is an inhibitor of type Q arylesterase activity
CAS Number:
5061-21-2
Molecular Weight:
164.99
Molecular Formula:
C4H5BrO2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Inhibit type Q arylesterase activity of the human serum paraoxonase PON1.
α-Bromo-γ-butyrolactone (CAS 5061-21-2) References
- Selective plasma hydrolysis of glucocorticoid gamma-lactones and cyclic carbonates by the enzyme paraoxonase: an ideal plasma inactivation mechanism. | Biggadike, K., et al. 2000. J Med Chem. 43: 19-21. PMID: 10633034
- Serum esterases. I. Two types of esterase (A and B) hydrolysing p-nitrophenyl acetate, propionate and butyrate, and a method for their determination. | ALDRIDGE, WN. 1953. Biochem J. 53: 110-7. PMID: 13032041
- Organocatalyzed ring-opening copolymerization of α-bromo-γ-butyrolactone with ε-caprolactone for the synthesis of functional aliphatic polyesters - pre-polymers for graft copolymerization. | Gao, C., et al. 2018. Des Monomers Polym. 21: 193-201. PMID: 30532666
- Biological Evaluation and Docking Studies of New Carbamate, Thiocarbamate, and Hydrazide Analogues of Acyl Homoserine Lactones as Vibrio fischeri-Quorum Sensing Modulators. | Zhang, Q., et al. 2020. Biomolecules. 10: PMID: 32183409
- A two-phase bromination process using tetraalkylammonium hydroxide for the practical synthesis of α-bromolactones from lactones. | Yamamoto, Y., et al. 2021. Beilstein J Org Chem. 17: 2906-2914. PMID: 34956409
- The Gln-Arg191 polymorphism of the human paraoxonase gene (HUMPONA) is not associated with the risk of coronary artery disease in Finns. | Antikainen, M., et al. 1996. J Clin Invest. 98: 883-5. PMID: 8770857
- Paraoxonase inhibits high-density lipoprotein oxidation and preserves its functions. A possible peroxidative role for paraoxonase. | Aviram, M., et al. 1998. J Clin Invest. 101: 1581-90. PMID: 9541487
- Establishing α-bromo-γ-butyrolactone as a platform for synthesis of functional aliphatic polyesters–bridging the gap between ROP and SET-LRP. | Olsén, P., Undin, J., Odelius, K., & Albertsson, A. C. 2014. Polymer Chemistry. 5(12): 3847-3854.
- Synthesis of polystyrene with cyclic, ionized and neutralized end groups and the self-assemblies templated by breath figures | Zhu, L. W., Yang, W., Ou, Y., Wan, L. S., & Xu, Z. K. 2014. Polymer Chemistry. 5(11): 3666-3672.
- Controlled copolymerization of the functional 5-membered lactone monomer, α-bromo-γ-butyrolactone, via selective organocatalysis | Undin, J., Olsén, P., Godfrey, J., Odelius, K., & Albertsson, A. C. 2016. Polymer. 87: 17-25.
- Study of 1, 2, 4-triazole-3 (5)-thiol Behavior in Reactions with 1-phenyl-1 H-pyrrole-2, 5-dione Derivatives and 3-bromodihydrofuran-2 (3 H)-one and Antimicrobial Activity of Products | Holota, S., Derkach, H., Antoniv, O., Slyvka, N., Kutsyk, R., Gzella, A., & Lesyk, R. 2020. Chemistry Proceedings. 3(1): 68.
- Mizoroki–Heck type reactions and synthesis of 1, 4-dicarbonyl compounds by heterogeneous organic semiconductor photocatalysis | Khamrai, J., Das, S., Savateev, A., Antonietti, M., & König, B. 2021. Green Chemistry. 23(5): 2017-2024.
- Synthesis of new 1, 4‐and 1, 5‐disubstituted N‐ethyl acetate and N‐α‐butyro‐γ‐lactone alkylimidazole derivatives as N‐acylhomoserine lactone analogs | Zhang, Q., Li, S. Z., Queneau, Y., & Soulère, L. 2021. Journal of Heterocyclic Chemistry. 58(12): 2298-2303.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
α-Bromo-γ-butyrolactone, 100 g | sc-219468 | 100 g | $1801.00 |
