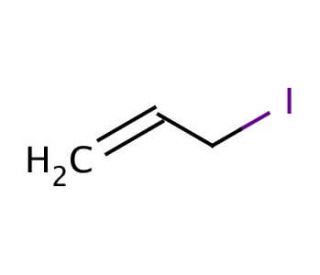
Molecular structure of Allyl iodide, CAS Number: 556-56-9
Allyl iodide (CAS 556-56-9)
Alternate Names:
3-Iodo-1-propene
CAS Number:
556-56-9
Molecular Weight:
167.98
Molecular Formula:
C3H5I
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Allyl iodide is used as a reagent in organic synthesis, particularly in the preparation of allyl ethers and esters. Allyl iodide has the ability to undergo nucleophilic substitution reactions, making it useful in the modification of organic molecules. In biochemistry, allyl iodide is used to introduce allyl groups into various compounds, allowing for the synthesis of complex molecules with specific properties. Its reactivity and selectivity in organic reactions make it a useful compound for the modification of biomolecules and the study of biochemical pathways.
Allyl iodide (CAS 556-56-9) References
- Ultraviolet photochemistry of trichlorovinylsilane and allyltrichlorosilane: vinyl radical (HCCH2) and allyl radical (H2CCHCH2) production in 193 nm photolysis. | DeSain, JD., et al. 2006. Phys Chem Chem Phys. 8: 2240-8. PMID: 16688306
- Shock wave study on the thermal unimolecular decomposition of allyl radicals. | Fernandes, RX., et al. 2005. J Phys Chem A. 109: 1063-70. PMID: 16833415
- The stability of allyl radicals following the photodissociation of allyl iodide at 193 nm. | Fan, H. and Pratt, ST. 2006. J Chem Phys. 125: 144302. PMID: 17042585
- Cis double allylation of cyclopropenes using cyclopropylindium reagents. | Hirashita, T., et al. 2007. Org Biomol Chem. 5: 2154-8. PMID: 17581660
- Letter: Silver mediated ester bond formation in the gas phase: substrate structure is important. | Khairallah, GN., et al. 2007. Eur J Mass Spectrom (Chichester). 13: 367-72. PMID: 18192731
- Oxidation state, aggregation, and heterolytic dissociation of allyl indium reagents. | Koszinowski, K. 2010. J Am Chem Soc. 132: 6032-40. PMID: 20377261
- N-Cyclo-hexyl-N-(prop-2-en-1-yl)benzene-sulfonamide. | Khan, IU., et al. 2009. Acta Crystallogr Sect E Struct Rep Online. 65: o3102. PMID: 21578829
- Gas-phase reactivity of group 11 dimethylmetallates with allyl iodide. | Rijs, NJ., et al. 2012. J Am Chem Soc. 134: 2569-80. PMID: 22280329
- Single pulse shock tube study of allyl radical recombination. | Fridlyand, A., et al. 2013. J Phys Chem A. 117: 4762-76. PMID: 23679206
- Gas-phase tyrosine-to-cysteine radical migration in model systems. | Lesslie, M., et al. 2015. Eur J Mass Spectrom (Chichester). 21: 589-97. PMID: 26307738
- Transition state region in the A-Band photodissociation of allyl iodide--A femtosecond extreme ultraviolet transient absorption study. | Bhattacherjee, A., et al. 2016. J Chem Phys. 144: 124311. PMID: 27036452
- Kinetics of the a-C3H5 + O2 reaction, investigated by photoionization using synchrotron radiation. | Schleier, D., et al. 2018. Phys Chem Chem Phys. 20: 10721-10731. PMID: 29340384
- Magnesium Bisamide-Mediated Halogen Dance of Bromothiophenes. | Yamane, Y., et al. 2018. Org Lett. 20: 1688-1691. PMID: 29513544
- Optimized synthesis and antiproliferative activity of desTHPdactylolides. | Chen, G., et al. 2018. Bioorg Med Chem. 26: 3514-3520. PMID: 29784275
- One-pot three component synthesis of 5-allyl-1,2,3-triazoles using copper(i) acetylides. | Reddy, PR., et al. 2018. RSC Adv. 8: 2759-2767. PMID: 35541454
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Allyl iodide, 25 g | sc-252360 | 25 g | $51.00 |
