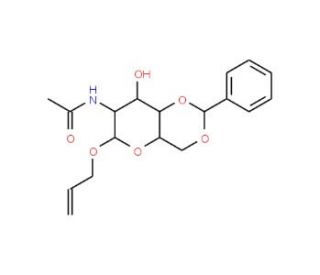
Allyl 2-(Acetylamino)-2-deoxy-4,6-O-(phenylmethylene)-α-D-glucopyranoside (CAS 63064-49-3)
QUICK LINKS
Allyl 2-(Acetylamino)-2-deoxy-4,6-O-(phenylmethylene)-α-D-glucopyranoside, a versatile chemical compound, has garnered considerable attention in carbohydrate chemistry research due to its unique structure and diverse applications. Its mechanism of action primarily revolves around its role as a glycosyl donor in the synthesis of complex carbohydrates and glycoconjugates. Through glycosylation reactions, this compound facilitates the formation of glycosidic bonds between sugar residues, enabling the construction of oligosaccharides with precise stereochemistry and linkage patterns. Researchers have extensively utilized Allyl 2-(Acetylamino)-2-deoxy-4,6-O-(phenylmethylene)-α-D-glucopyranoside in the synthesis of carbohydrate-based molecules, including glycopeptides, glycolipids, and natural product analogs, for structure-activity relationship studies and biological evaluations. Additionally, its use as a glycosyl donor in enzymatic glycosylation reactions has been explored, offering an enzymatic alternative for synthesizing complex carbohydrates with high efficiency and selectivity. Moreover, this compound serves as a valuable building block for the preparation of carbohydrate-modified materials, such as glycosylated polymers, nanoparticles, and surfaces, which find applications in various fields, including biomaterials science, drug delivery, and diagnostics. Its ability to undergo chemical modification further enhances its utility in the design and synthesis of diverse carbohydrate derivatives for investigating carbohydrate-protein interactions, elucidating carbohydrate-mediated biological processes, and developing innovative carbohydrate-based materials for biomedical and biotechnological applications.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Allyl 2-(Acetylamino)-2-deoxy-4,6-O-(phenylmethylene)-α-D-glucopyranoside, 100 mg | sc-210783 | 100 mg | $320.00 |
