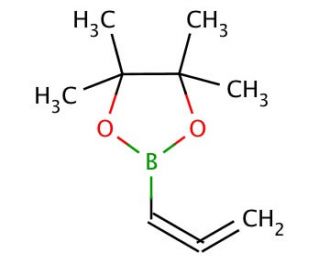
Allenylboronic acid pinacol ester (CAS 865350-17-0)
QUICK LINKS
Allenylboronic acid pinacol ester is a versatile compound widely utilized in organic synthesis and chemical research for its ability to partake in cross-coupling reactions, serving as a precursor for the synthesis of complex molecular structures. This compound is especially valued for its role in Suzuki-Miyaura coupling reactions, where it acts as an organoboron reagent to form carbon-carbon bonds with aryl or vinyl halides under palladium catalysis. The presence of the allenyl group in the molecule allows for the formation of highly conjugated systems through subsequent transformations, enabling researchers to construct architecturally complex molecules with precision. The pinacol ester moiety in its structure enhances its stability and solubility, facilitating its handling and application in various reaction conditions. Its utilization in chemical synthesis is pivotal for the development of novel materials, catalysts, and synthesis pathways, reflecting its significance in advancing the field of organic chemistry.
Allenylboronic acid pinacol ester (CAS 865350-17-0) References
- Palladium-catalyzed annulation reactions for diastereoselective cyclopentene synthesis. | Kohn, BL. and Jarvo, ER. 2011. Org Lett. 13: 4858-61. PMID: 21854011
- Site- and enantioselective formation of allene-bearing tertiary or quaternary carbon stereogenic centers through NHC-Cu-catalyzed allylic substitution. | Jung, B. and Hoveyda, AH. 2012. J Am Chem Soc. 134: 1490-3. PMID: 22214185
- Understanding the mechanism of the asymmetric propargylation of aldehydes promoted by 1,1'-bi-2-naphthol-derived catalysts. | Grayson, MN. and Goodman, JM. 2013. J Am Chem Soc. 135: 6142-8. PMID: 23517191
- Silver-Catalyzed Enantioselective Propargylation Reactions of N-Sulfonylketimines. | Osborne, CA., et al. 2015. Org Lett. 17: 5340-3. PMID: 26506430
- Sixteen isostructural phosphonate metal-organic frameworks with controlled Lewis acidity and chemical stability for asymmetric catalysis. | Chen, X., et al. 2017. Nat Commun. 8: 2171. PMID: 29259195
- N-Heterocyclic Carbene-Cu-Catalyzed Enantioselective Allenyl Conjugate Addition. | Chong, Q., et al. 2018. Org Lett. 20: 6896-6900. PMID: 30350670
- Efficient method for propargylation of aldehydes promoted by allenylboron compounds under microwave irradiation. | Freitas, JJR., et al. 2020. Beilstein J Org Chem. 16: 168-174. PMID: 32117473
- Allenylboronic Acid Pinacol Ester: A Selective Partner for [4 + 2] Cycloadditions. | Labadie, N., et al. 2021. Org Lett. 23: 5081-5085. PMID: 34151574
- Diels-Alder Reactivity of Allenylboronic Acid Pinacol Ester and Related Dienophiles: Mechanistic Studies and Distortion/Interaction-Activation Strain Model Analysis. | Labadie, N. and Pellegrinet, SC. 2022. J Org Chem. 87: 16776-16784. PMID: 36440689
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Allenylboronic acid pinacol ester, 1 g | sc-262980 | 1 g | $153.00 | |||
Allenylboronic acid pinacol ester, 5 g | sc-262980A | 5 g | $520.00 |
