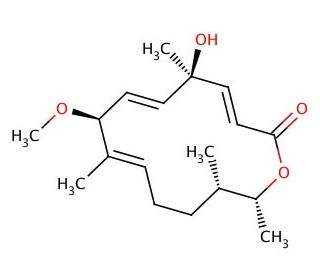
Albocycline (CAS 25129-91-3)
QUICK LINKS
Albocycline is a macrolide antibiotic identified from the bacterium Streptomyces species, noted for its complex macrolide structure. Although primarily categorized within antibiotics, albocycline′s structure and mechanisms have been studied, particularly within organic chemistry and microbial ecology. The structural composition of albocycline includes a large macrocyclic lactone ring which is crucial for its interaction with other molecules. In research contexts, one of the primary mechanisms explored is albocycline′s ability to bind to bacterial ribosomes in a non-inhibitory manner. This binding study provides insights into the interactions between macrolides and ribosomal structures, enriching understanding of molecular docking and binding dynamics in a biochemical setting. Furthermore, albocycline has been a subject in the study of natural product biosynthesis. Researchers investigate its biosynthetic pathways to comprehend the enzymatic processes that lead to the formation of complex macrolide structures. Such studies are pivotal in synthetic biology and bioengineering, where insights gained can facilitate the development of methods to synthesize similar structures via engineered microbial pathways.
Albocycline (CAS 25129-91-3) References
- Elucidating the inhibition of peptidoglycan biosynthesis in Staphylococcus aureus by albocycline, a macrolactone isolated from Streptomyces maizeus. | Liang, H., et al. 2018. Bioorg Med Chem. 26: 3453-3460. PMID: 29805074
- Albocycline-type Macrolides with Antibacterial Activities from Streptomyces sp. 4205. | Gu, CZ., et al. 2019. Chem Biodivers. 16: e1800344. PMID: 30358064
- Staphylococcus aureus resistance to albocycline can be achieved by mutations that alter cellular NAD/PH pools. | Scherzi, T., et al. 2021. Bioorg Med Chem. 32: 115995. PMID: 33477021
- Albocycline Is the Main Bioactive Antifungal Compound Produced by Streptomyces sp. OR6 against Verticillium dahliae. | Calvo-Peña, C., et al. 2023. Plants (Basel). 12: PMID: 37896074
- Mode of action of albocycline, an inhibitor of nicotinate biosynthesis. | Reusser, F. 1969. J Bacteriol. 100: 11-3. PMID: 4241976
- Studies on a new antibiotic, albocycline. II. Taxonomic studies on albocycline-producing strains. | Furumai, T., et al. 1968. J Antibiot (Tokyo). 21: 85-90. PMID: 5674371
- Studies on an antibiotic, albocycline VII. Minor components of albocycline. | Harada, K., et al. 1984. J Antibiot (Tokyo). 37: 1187-97. PMID: 6501089
- Biomodification of albocycline by Streptomyces venezuelae. | Slechta, L., et al. 1978. J Antibiot (Tokyo). 31: 319-23. PMID: 659330
- Albocycline: structure determination by x-ray crystallography. | Thomas, RC. and Chidester, CG. 1982. J Antibiot (Tokyo). 35: 1658-64. PMID: 7166531
- Biosynthesis of albocycline: origin of the carbon skeleton. | Taddei, A. and Zeeck, A. 1997. J Antibiot (Tokyo). 50: 526-8. PMID: 9268011
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Albocycline, 1 mg | sc-396530 | 1 mg | $127.00 | |||
Albocycline, 5 mg | sc-396530A | 5 mg | $440.00 |
