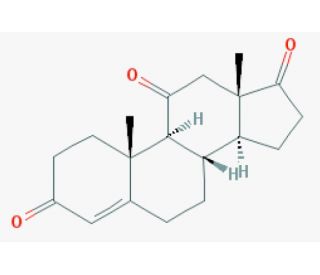
Molecular structure of Adrenosterone, CAS Number: 382-45-6
Adrenosterone (CAS 382-45-6)
Alternate Names:
11-Ketoandrostenedione; 4-Androstene-3,11,17-trione; Androst-4-ene-3;11,17-trione; Reichstein′s substance G
CAS Number:
382-45-6
Purity:
≥97%
Molecular Weight:
300.39
Molecular Formula:
C19H24O3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Adrenosterone is an endogenous steroid hormone and belongs to the group of glucocorticoids. It is as a precursor for cortisol. Adrenosterone is a strong mineralocorticoid and contributes to the regulation of blood pressure, electrolyte balance, and water balance within the body.
Adrenosterone (CAS 382-45-6) References
- Biotransformation of adrenosterone by filamentous fungus, Cunninghamella elegans. | Choudhary, MI., et al. 2007. Steroids. 72: 923-9. PMID: 17889091
- Urinary metabonomics study on biochemical changes in an experimental model of chronic renal failure by adenine based on UPLC Q-TOF/MS. | Zhao, YY., et al. 2012. Clin Chim Acta. 413: 642-9. PMID: 22227165
- Multiple mechanisms of ligand interaction with the human organic cation transporter, OCT2. | Harper, JN. and Wright, SH. 2013. Am J Physiol Renal Physiol. 304: F56-67. PMID: 23034939
- Tandem mass spectrometry approach for the investigation of the steroidal metabolism: structure-fragmentation relationship (SFR) in anabolic steroids and their metabolites by ESI-MS/MS analysis. | Musharraf, SG., et al. 2013. Steroids. 78: 171-81. PMID: 23159734
- Total synthesis of starfish saponin goniopectenoside B. | Xiao, G. and Yu, B. 2013. Chemistry. 19: 7708-12. PMID: 23649953
- Pathways and genes involved in steroid hormone metabolism in male pigs: a review and update. | Robic, A., et al. 2014. J Steroid Biochem Mol Biol. 140: 44-55. PMID: 24239507
- Regio- and stereospecific hydroxylation of various steroids at the 16α position of the D ring by the Streptomyces griseus cytochrome P450 CYP154C3. | Makino, T., et al. 2014. Appl Environ Microbiol. 80: 1371-9. PMID: 24334658
- Isaria fumosorosea KCh J2 Entomopathogenic Strain as an Effective Biocatalyst for Steroid Compound Transformations. | Kozłowska, E., et al. 2017. Molecules. 22: PMID: 28891949
- Hydroxylation of Steroids by a Microbial Substrate-Promiscuous P450 Cytochrome (CYP105D7): Key Arginine Residues for Rational Design. | Ma, B., et al. 2019. Appl Environ Microbiol. 85: PMID: 31540985
- A Novel Zebrafish Model of Metastasis Identifies the HSD11β1 Inhibitor Adrenosterone as a Suppressor of Epithelial-Mesenchymal Transition and Metastatic Dissemination. | Nakayama, J., et al. 2020. Mol Cancer Res. 18: 477-487. PMID: 31748280
- 17β-Hydroxysteroid dehydrogenase type 12 is responsible for maturation-inducing steroid synthesis during oocyte maturation in Nile tilapia. | Aranyakanont, C., et al. 2020. Gen Comp Endocrinol. 290: 113399. PMID: 31982399
- Xmrks the Spot: Fish Models for Investigating Epidermal Growth Factor Receptor Signaling in Cancer Research. | Monroe, JD., et al. 2021. Cells. 10: PMID: 34067095
- Spontaneous conversion of prenyl halides to acids: application in metal-free preparation of deuterated compounds under mild conditions. | Darshana, D., et al. 2021. Org Biomol Chem. 19: 7390-7402. PMID: 34296730
- Gastrulation Screening to Identify Anti-metastasis Drugs in Zebrafish Embryos. | Nakayama, J., et al. 2022. Bio Protoc. 12: PMID: 36313195
- Development of mass spectrometry-based methods for the detection of 11-ketotestosterone and 11-ketodihydrotestosterone. | Piper, T. and Thevis, M. 2023. Drug Test Anal.. PMID: 36652270
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Adrenosterone, 5 g | sc-233824 | 5 g | $260.00 | |||
Adrenosterone, 25 g | sc-233824A | 25 g | $1200.00 | |||
Adrenosterone, 100 g | sc-233824B | 100 g | $4000.00 |
