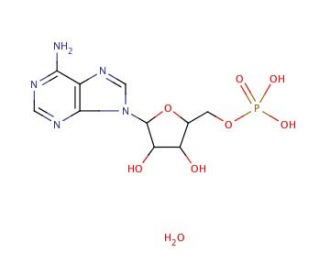
Adenosine 5′-monophosphate monohydrate (CAS 18422-05-4)
QUICK LINKS
Adenosine 5′-monophosphate (AMP) monohydrate is a nucleotide present in all living cells, comprised of a ribose sugar, a phosphate group, and an adenine base. Its significance lies in its integral role within various biochemical pathways such as energy metabolism, gene expression, and signal transduction. This compound, known as Adenosine 5′-monophosphate monohydrate, holds diverse research applications, both within living organisms and controlled environments. Within organisms, it serves to delve into cell signaling pathways, gene expression, and energy metabolism. Acting as an allosteric enzyme activator, Adenosine 5′-monophosphate monohydrate participates in the regulation of key biochemical routes. By binding to specific regulatory proteins, it influences the activation or inhibition of target enzymes. This mode of operation is pivotal in governing energy metabolism, gene expression, and signal transduction pathways.
Adenosine 5′-monophosphate monohydrate (CAS 18422-05-4) References
- Counter-current chromatographic separation of nucleic acid constituents with a hydrophilic solvent system. | Shibusawa, Y., et al. 2010. J Chromatogr A. 1217: 3457-60. PMID: 20362294
- Increasing surface enhanced Raman spectroscopy effect of RNA and DNA components by changing the pH of silver colloidal suspensions. | Primera-Pedrozo, OM., et al. 2012. Spectrochim Acta A Mol Biomol Spectrosc. 87: 77-85. PMID: 22169024
- Transport of the coumarin metabolite 7-hydroxycoumarin glucuronide is mediated via multidrug resistance-associated proteins 3 and 4. | Wittgen, HG., et al. 2012. Drug Metab Dispos. 40: 1076-9. PMID: 22415933
- Separation of nucleotides by hydrophilic interaction chromatography using the FRULIC-N column. | Padivitage, NL., et al. 2013. Anal Bioanal Chem. 405: 8837-48. PMID: 23995506
- Infrared multiple photon dissociation action spectroscopy of deprotonated RNA mononucleotides: gas-phase conformations and energetics. | Nei, YW., et al. 2013. J Phys Chem A. 117: 10634-49. PMID: 24053133
- Counter-Current Chromatographic Separation of Nucleic Acid Constituents with a Polar Volatile Organic-Aqueous Two-Phase Solvent Systems with ELSD Detection. | Shibusawa, Y., et al. 2012. Open Anal Chem J. 6: 9-14. PMID: 24634703
- Controlling the complexation of polysaccharides into multi-functional colloidal assemblies for nanomedicine. | Costalat, M., et al. 2014. J Colloid Interface Sci. 430: 147-56. PMID: 24998067
- Simultaneous determination of total dissolved nitrogen and total dissolved phosphorus in natural waters with an on-line UV and thermal digestion. | Lin, K., et al. 2018. Talanta. 185: 419-426. PMID: 29759221
- Hydrophilic interaction liquid chromatography coupled with quadrupole-orbitrap ultra high resolution mass spectrometry to quantitate nucleobases, nucleosides, and nucleotides during white tea withering process. | Zhao, F., et al. 2018. Food Chem. 266: 343-349. PMID: 30381196
- Non-targeted metabolomics unravels a media-dependent prodiginines production pathway in Streptomyces coelicolor A3(2). | Lim, Y., et al. 2018. PLoS One. 13: e0207541. PMID: 30485320
- Novel adsorptive materials by adenosine 5'-triphosphate imprinted-polymer over the surface of polystyrene nanospheres for selective separation of adenosine 5'-triphosphate biomarker from urine. | Jadda, R., et al. 2019. J Sep Sci. 42: 3662-3678. PMID: 31591808
- Hydrophilic molecularly imprinted polymers functionalized magnetic carbon nanotubes for selective extraction of cyclic adenosine monophosphate from winter jujube. | Li, F., et al. 2021. J Sep Sci. 44: 2131-2142. PMID: 33721391
- Simultaneous oxidation and analysis of TOC-TN-TP in one pot reactor. | Shin, D., et al. 2022. Chemosphere. 292: 133336. PMID: 34922955
- Synthetic torpor protects rats from exposure to accelerated heavy ions. | Puspitasari, A., et al. 2022. Sci Rep. 12: 16405. PMID: 36180516
- Potent inhibitors of human inosine monophosphate dehydrogenase type II. Fluorine-substituted analogues of thiazole-4-carboxamide adenine dinucleotide. | Zatorski, A., et al. 1995. J Med Chem. 38: 1098-105. PMID: 7707313
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Adenosine 5′-monophosphate monohydrate, 5 g | sc-214503 | 5 g | $64.00 | |||
Adenosine 5′-monophosphate monohydrate, 25 g | sc-214503A | 25 g | $233.00 | |||
Adenosine 5′-monophosphate monohydrate, 250 g | sc-214503B | 250 g | $357.00 | |||
Adenosine 5′-monophosphate monohydrate, 1 kg | sc-214503C | 1 kg | $734.00 | |||
Adenosine 5′-monophosphate monohydrate, 5 kg | sc-214503D | 5 kg | $2396.00 |
