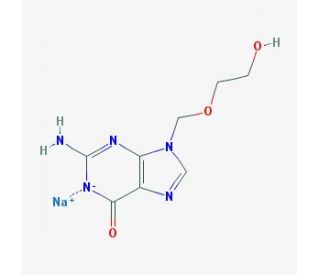
Molecular structure of Acyclovir sodium, CAS Number: 69657-51-8
Acyclovir sodium (CAS 69657-51-8)
Application:
Acyclovir sodium is a guanosine analog for biochemical research
CAS Number:
69657-51-8
Purity:
≥98%
Molecular Weight:
247.19
Molecular Formula:
C8H10N5O3•Na
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Acyclovir sodium is a nucleoside analog that functions as an antiviral agent. It inhibits the replication of herpes simplex virus by interfering with the viral DNA polymerase. Acyclovir sodium is phosphorylated by viral thymidine kinase to its active form, which then competes with deoxyguanosine triphosphate for incorporation into viral DNA. This results in chain termination, preventing further viral DNA synthesis. Acyclovir sodium specifically targets infected cells, as it requires viral thymidine kinase for activation. By disrupting viral DNA replication, Acyclovir sodium effectively limits the spread and proliferation of the herpes simplex virus within the host cell.
Acyclovir sodium (CAS 69657-51-8) References
- Design and development of multivesicular liposomal depot delivery system for controlled systemic delivery of acyclovir sodium. | Jain, SK., et al. 2005. AAPS PharmSciTech. 6: E35-41. PMID: 16353961
- Natural history and treatment of varicella-zoster in high-risk populations. | Gnann, JW. and Whitley, RJ. 1991. J Hosp Infect. 18 Suppl A: 317-29. PMID: 1679798
- Enhanced transdermal delivery of acyclovir sodium via elastic liposomes. | Jain, SK., et al. 2008. Drug Deliv. 15: 141-7. PMID: 18379926
- Stability of acyclovir sodium after reconstitution in 0.9% sodium chloride inection and storage in polypropylene syringes for pediatric use. | Ling, J. and Gupta, VD. 2001. Int J Pharm Compd. 5: 75-7. PMID: 23981803
- Oral acyclovir reduces the incidence of recurrent herpes simplex keratitis in rabbits after penetrating keratoplasty. | Beyer, CF., et al. 1989. Arch Ophthalmol. 107: 1200-5. PMID: 2547352
- Relapse of herpes simplex encephalitis after conventional acyclovir therapy. | VanLandingham, KE., et al. 1988. JAMA. 259: 1051-3. PMID: 3339802
- Drug-use criteria for acyclovir sodium injection. | Melamed, AJ. and Brown, AE. 1988. Am J Hosp Pharm. 45: 382-3. PMID: 3364439
- Varicella pneumonia. | Schlossberg, D. and Littman, M. 1988. Arch Intern Med. 148: 1630-2. PMID: 3382308
- Visual compatibility of acyclovir sodium with commonly used intravenous drugs simulated Y-site injection. | Forman, JK., et al. 1987. Am J Hosp Pharm. 44: 1408-9. PMID: 3618621
- Fabrication of Stimuli-Responsive Quince/Mucin Co-Poly (Methacrylate) Hydrogel Matrices for the Controlled Delivery of Acyclovir Sodium: Design, Characterization and Toxicity Evaluation. | Aslam, A., et al. 2023. Pharmaceutics. 15: PMID: 36839971
- Antiviral agents. | Hermans, PE. and Cockerill, FR. 1983. Mayo Clin Proc. 58: 217-22. PMID: 6339831
- Failure of acyclovir sodium therapy in herpes simplex encephalitis. | Gürses, N., et al. 1996. Ann Trop Paediatr. 16: 173-5. PMID: 8790683
- Stability of acyclovir sodium 1, 7, and 10 mg/mL in 5% dextrose injection and 0.9% sodium chloride injection. | Zhang, Y., et al. 1998. Am J Health Syst Pharm. 55: 574-7. PMID: 9544334
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Acyclovir sodium, 1 g | sc-352990 | 1 g | $140.00 | |||
Acyclovir sodium, 25 g | sc-352990A | 25 g | $555.00 |
