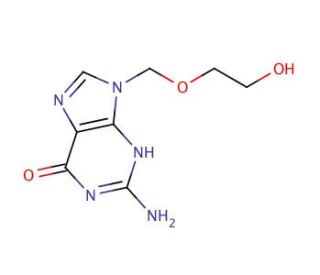
Acyclovir (CAS 59277-89-3)
See product citations (2)
QUICK LINKS
Acyclovir, a synthetic nucleoside analogue, primarily functions by inhibiting viral DNA synthesis, showcasing a unique mechanism of action that has been extensively utilized in virological research. Its molecular structure mimics that of guanine nucleosides, allowing it to be selectively incorporated into viral DNA by viral DNA polymerase. Upon integration, acyclovir acts as a chain terminator. Unlike normal DNA replication, where the chain continues to elongate, the incorporation of acyclovir halts this process, effectively preventing the virus from replicating its genetic material. This specific interaction has made acyclovir a focal point in studies exploring the replication mechanisms of herpes simplex virus (HSV) and varicella-zoster virus (VZV), among others. Its ability to selectively target viral over host polymerases underscores its utility in research aimed at understanding viral pathogenesis and developing strategies for antiviral intervention. Through elucidating the dynamics of viral replication and the host′s cellular response, acyclovir has contributed significantly to the field of virology, offering insights that extend beyond its direct application, influencing the design and synthesis of future antiviral agents.
Acyclovir (CAS 59277-89-3) References
- Samarangenin B from Limonium sinense suppresses herpes simplex virus type 1 replication in Vero cells by regulation of viral macromolecular synthesis. | Kuo, YC., et al. 2002. Antimicrob Agents Chemother. 46: 2854-64. PMID: 12183238
- Acyclovir: a decade later. | Whitley, RJ. and Gnann, JW. 1992. N Engl J Med. 327: 782-9. PMID: 1288525
- Murine cytomegalovirus DNA polymerase: purification, characterization and role in the antiviral activity of acyclovir. | Ochiai, H., et al. 1992. Antiviral Res. 17: 1-16. PMID: 1310580
- Selective inhibition of proliferation in v-abl- and bcr-abl-transformed cells by a nucleoside analog. | Cioé, L., et al. 1992. J Biol Chem. 267: 22178-82. PMID: 1331046
- Rapid determination of acyclovir in plasma and cerebrospinal fluid by micellar electrokinetic chromatography with direct sample injection and its clinical application. | Yeh, HH., et al. 2006. Electrophoresis. 27: 819-26. PMID: 16411274
- Oral bioavailability enhancement of acyclovir by self-microemulsifying drug delivery systems (SMEDDS). | Patel, D. and Sawant, KK. 2007. Drug Dev Ind Pharm. 33: 1318-26. PMID: 18097805
- Valacyclovir and acyclovir pharmacokinetics in immunocompromised children. | Bomgaars, L., et al. 2008. Pediatr Blood Cancer. 51: 504-8. PMID: 18561175
- Controlled ex-vivo plasma hydrolysis of valaciclovir to acyclovir demonstration using tandem mass spectrometry. | Goswami, D., et al. 2011. Biomed Chromatogr. 25: 1189-200. PMID: 21400550
- History, pharmacokinetics, and pharmacology of acyclovir. | King, DH. 1988. J Am Acad Dermatol. 18: 176-9. PMID: 2828440
- Enhanced acyclovir delivery using w/o type microemulsion: preclinical assessment of antiviral activity using murine model of zosteriform cutaneous HSV-1 infection. | Kaur, A., et al. 2018. Artif Cells Nanomed Biotechnol. 46: 346-354. PMID: 28403666
- In vitro susceptibility of cytomegalovirus isolates from immunocompromised patients to acyclovir and ganciclovir. | Cole, NL. and Balfour, HH. 1987. Diagn Microbiol Infect Dis. 6: 255-61. PMID: 3032503
- Acyclovir Unresponsive Herpes Simplex Encephalitis in a child successfully treated with the addition of Foscarnet: Case report. | Gayretli Aydin, ZG., et al. 2019. Arch Argent Pediatr. 117: e47-e51. PMID: 30652455
- Inhibitory effect of a nucleoside analog, acyclovir, on leukemia cells. | Nishimaki, J., et al. 1996. Leuk Res. 20: 415-20. PMID: 8683981
- Improvement of gene therapy for ovarian cancer by using acyclovir instead of ganciclovir in adenovirus mediated thymidine kinase gene therapy. | Tong, XW., et al. 1998. Anticancer Res. 18: 713-8. PMID: 9615710
- Superior cytotoxicity with ganciclovir compared with acyclovir and 1-beta-D-arabinofuranosylthymine in herpes simplex virus-thymidine kinase-expressing cells: a novel paradigm for cell killing. | Rubsam, LZ., et al. 1998. Cancer Res. 58: 3873-82. PMID: 9731497
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Acyclovir, 50 mg | sc-202906 | 50 mg | $150.00 | |||
Acyclovir, 500 mg | sc-202906A | 500 mg | $940.00 |
