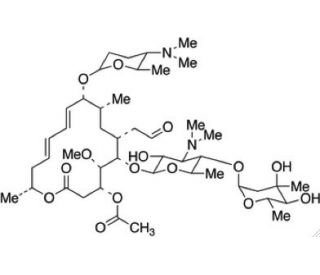
Acetyl Spiramycin (CAS 24916-51-6)
QUICK LINKS
Acetyl Spiramycin refers to a chemically acetylated derivative of Spiramycin. It effectively combats various bacterial infections, and is referred to as spiramycin, spiramycin acetate, or spiramycin I. It′s mechanism of action involves hindering bacterial growth and replication by obstructing protein synthesis. It achieves this by binding to the 50S ribosomal subunit enzyme, preventing peptide bond formation, ultimately impeding bacterial proliferation. This compound is studied for its distinct biochemical properties and interactions, especially in the context of its modified efficacy and stability compared to its parent compound. The acetylation potentially affects its solubility, distribution, and the mechanism of interaction with bacterial targets, making it a subject of interest in biochemical and pharmaceutical research to understand its behavior and applications in various contexts.
Acetyl Spiramycin (CAS 24916-51-6) References
- Streptococcus pyogenes resistance to erythromycin in relation to macrolide consumption in Spain (1986-1997). | Granizo, JJ., et al. 2000. J Antimicrob Chemother. 46: 959-64. PMID: 11102415
- No significant increase in chromosome aberrations and sister chromatid exchanges in cultured human lymphocytes treated with spiramycin. | Rencüzoğullari, E., et al. 2002. Teratog Carcinog Mutagen. 22: 51-8. PMID: 11754387
- Control of fed-batch fermentations. | Lee, J., et al. 1999. Biotechnol Adv. 17: 29-48. PMID: 14538142
- Antibacterial macrolides: a drug class with a complex pharmacological profile. | Abu-Gharbieh, E., et al. 2004. Pharmacol Res. 50: 211-22. PMID: 15225662
- Antimicrobial susceptibility of 1042 strains of Streptococcus mutans and Streptococcus sobrinus: comparison from 1985 to 1989. | Liebana, J., et al. 1991. Oral Microbiol Immunol. 6: 146-50. PMID: 1945497
- Determination of 17 macrolide antibiotics and avermectins residues in meat with accelerated solvent extraction by liquid chromatography-tandem mass spectrometry. | Tao, Y., et al. 2012. J Chromatogr B Analyt Technol Biomed Life Sci. 897: 64-71. PMID: 22542398
- Serological Analysis and Drug Resistance of Chlamydia pneumoniae and Mycoplasma pneumoniae in 4500 Healthy Subjects in Shenzhen, China. | Du, D., et al. 2017. Biomed Res Int. 2017: 3120138. PMID: 29057257
- Research on the effect and mechanism of antimicrobial peptides HPRP-A1/A2 work against Toxoplasma gondii infection. | Liu, R., et al. 2019. Parasite Immunol. 41: e12619. PMID: 30788848
- Annual report of the Women's Health Care Committee, Japan Society of Obstetrics and Gynecology, 2019. | Kitawaki, J. and Kato, K. 2020. J Obstet Gynaecol Res. 46: 39-48. PMID: 31595599
- Targeted analysis of six emerging derivatives or metabolites together with 25 common macrolides in milk using Quick, Easy, Cheap, Effective, Rugged and Safe extraction and ultra-performance liquid chromatography quadrupole/electrostaticfield orbitrap mass spectrometry. | Wang, J., et al. 2020. J Sep Sci. 43: 3719-3734. PMID: 32725879
- [Experimental study on the antistaphylococcal activity of acetyl-spiramycin]. | Kawamori, Y., et al. 1967. J Antibiot B. 20: 10-1. PMID: 5299296
- Region fuzzy control for batch processes Part 2. Feed timing prediction and control for an antibiotic fermentation production process | FU, C. S., WANG, S. Q., & WANG, J. C. 1990. International journal of systems science. 21(10): 1911-1921.
- In Vivo Genotoxic Effects of Spiramycin in Rat Bone Marrow Cells | HB, I., & Topaktas, M. 1999. Cytologia. 64(3): 277-283.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Acetyl Spiramycin, 50 mg | sc-217568 | 50 mg | $367.00 |
