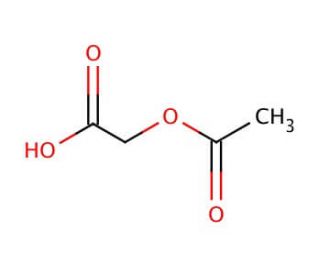
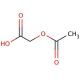
Acetoxyacetic acid (CAS 13831-30-6)
QUICK LINKS
Acetoxyacetic acid is a chemical compound that functions as an acetylating agent in various organic synthesis reactions. Its mechanism of action involves the transfer of an acetyl group to other molecules, leading to the formation of acetoxyacetate derivatives. Acetoxyacetic Acid may play a role in the acetylation of alcohols, phenols, and amines, facilitating the introduction of acetyl groups into these compounds. Acetoxyacetic acid is utilized for its ability to modify Acetoxyacetic acid structure of organic molecules, allowing for the synthesis of new compounds with specific properties. Its mechanism of action involves the acetylation of functional groups, enabling the creation of diverse chemical derivatives for experimental purposes. Acetoxyacetic Acid′s function as an acetylating agent contributes to the modification and diversification of organic molecules, supporting the exploration of novel chemical structures in development applications.
Acetoxyacetic acid (CAS 13831-30-6) References
- Ultrasonic chemical oxidative degradations of 1,3-dialkylimidazolium ionic liquids and their mechanistic elucidations. | Li, X., et al. 2007. Dalton Trans. 1875-80. PMID: 17702165
- Unusual ent-atisane type diterpenoids with 2-oxopropyl skeleton from the roots of Euphorbia ebracteolata and their antiviral activity against human rhinovirus 3 and enterovirus 71. | Wang, B., et al. 2018. Bioorg Chem. 81: 234-240. PMID: 30153588
- The deactivation of rabbit neutrophils by chemotactic factor and the nature of the activatable esterase. | Ward, PA. and Becker, EL. 1968. J Exp Med. 127: 693-709. PMID: 5642465
- Dibenz[b,e]oxepinalkanoic acids as nonsteroidal antiinflammatory agents. 4. Synthesis and evaluation of 4-(4,10-dihydro-10-oxothieno[3,2-c] [1]benzoxepin-8-yl)butanol and -butyric acid and related derivatives. | Martin, LL., et al. 1984. J Med Chem. 27: 372-6. PMID: 6607999
- Determination of stability constants from diffusion coefficient data | , et al. (1985). Proceedings of the Indian Academy of Sciences - Chemical Sciences. volume 95,: pages 517–524.
- Use of tellurium(IV) and tellurium(VI) as oxidants in organic synthesis | Jan Bergman and Lars Engman. 1982,. J. Org. Chem. 47, 26,: 5191–5196.
- Reactions of lead tetra-acetate. Part XIV. The base-catalysed decomposition of lead tetra-acetate in acetic acid | J. Chem. Soc. B,. 1968,. Journal of the Chemical Society B: Physical Organic. 781-784.
- Decomposition of acetyl peroxide in the presence of acetate salts | Tamas Koenig and R. Wielesek. 1969. J. Am. Chem. Soc., 91, 10,: 2551–2557.
- Thermolysis of manganese(III) acetate in acetic anhydride and in acetic anhydride/acetic acid mixtures | and W. J. de Klein. 1977. Recueil des Travaux Chimiques des Pays-Bas. Volume96, Issue1: Pages 22-25.
- Synthesis of thiophene-containing steroid-like molecules via olefinic cyclization reactions | and A. Corvers, J. H. van Mil, M. M. E. Sap, H. M. Buck. 1977. Recueil des Travaux Chimiques des Pays-Bas. Volume96, Issue1: Pages 18-22.
- Kinetics and mechanism of the gas-phase elimination of primary, secondary and tertiary 2-acetoxycarboxylic acids | and Gabriel Chuchani, Rosa M Dominguez, Armando Herize, Roman Romero. November 2000. Journal of Physical Organic Chemistry. Volume13, Issue11: Pages 757-764.
- Valuable oxygenates by aerobic oxidation of polymers using metal/bromide homogeneous catalysts | W Partenheimer - Catalysis today, 2003 - Elsevier. 15 June 2003,. Catalysis Today. Volume 81, Issue 2,: Pages 117-135.
- Synthesis of 2,5-Diformylfuran and Furan-2,5-Dicarboxylic Acid by Catalytic Air-Oxidation of 5-Hydroxymethylfurfural. Unexpectedly Selective Aerobic Oxidation of Benzyl Alcohol to Benzaldehyde with Metal=Bromide Catalysts | and Walt Partenheimer, Vladimir V. Grushin. 29 January 2001. Advanced Synthesis & Catalysis. Volume343, Issue1: Pages 102-111.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Acetoxyacetic acid, 5 g | sc-227204 | 5 g | $53.00 |
