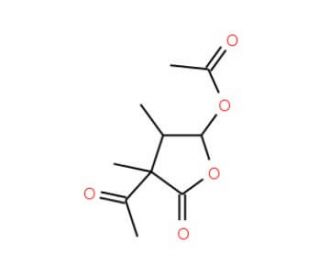
Acetomycin (CAS 510-18-9)
QUICK LINKS
Acetomycin, a chemical compound of interest in scientific research, has captivated researchers due to its unique properties and potential applications. This compound′s mechanism of action revolves around its ability to interfere with microbial metabolism, particularly by disrupting essential cellular processes. By targeting specific enzymatic pathways crucial for microbial growth and survival, acetomycin demonstrates potent antimicrobial activity against a diverse range of microorganisms. Acetomycin has been extensively explored for its antimicrobial efficacy and mechanism of action. Researchers have delved into its molecular interactions with microbial targets, unraveling the complex biochemical pathways affected by this compound. Insights gained from these studies have not only expanded our understanding of microbial physiology but have also paved the way for the development of novel antimicrobial agents. Furthermore, acetomycin′s antimicrobial properties have sparked interest in its potential applications. Researchers have investigated its role in environmental microbiology, exploring its impact on microbial communities and ecosystem dynamics. Additionally, acetomycin′s ability to selectively target specific microorganisms has led to its exploration in industrial microbiology, where it holds promise for applications such as food preservation and bioprocessing.
Acetomycin (CAS 510-18-9) References
- Total synthesis of (+/-)-acetomycin and design of esterase-resistant analogs. | Uenishi, J., et al. 1999. Chem Pharm Bull (Tokyo). 47: 517-23. PMID: 10319430
- Formation of chiral quaternary carbon stereocenters using silylene transfer reactions: enantioselective synthesis of (+)-5-epi-acetomycin. | Calad, SA. and Woerpel, KA. 2007. Org Lett. 9: 1037-40. PMID: 17305349
- The structure and absolute configuration of acetomycin. | Cano, FH., et al. 1988. Acta Crystallogr C. 44 (Pt 5): 919-21. PMID: 3271088
- Biological effects of acetomycin. I. Activity against tumor cells in vitro and in vivo. | Mamber, SW., et al. 1987. J Antibiot (Tokyo). 40: 73-6. PMID: 3558118
- Biological effects of acetomycin. II. Inactivation by esterases in vitro. | Mamber, SW., et al. 1987. J Antibiot (Tokyo). 40: 77-80. PMID: 3558119
- Identification of Acetomycin as an Antifungal Agent Produced by Termite Gut-Associated Streptomycetes against Pyrrhoderma noxium. | Adra, C., et al. 2024. Antibiotics (Basel). 13: PMID: 38247604
- The structure of acetomycin. Spectroscopic characterization and X-ray analysis of a bromo derivative. | Uhr, H., et al. 1985. J Antibiot (Tokyo). 38: 1684-90. PMID: 4093332
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Acetomycin, 1 mg | sc-202035 | 1 mg | $270.00 | |||
Acetomycin, 5 mg | sc-202035A | 5 mg | $772.00 |
