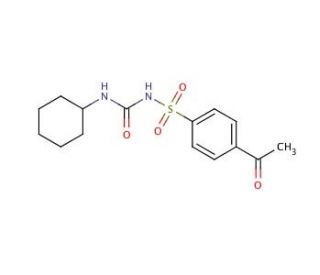
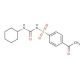
Acetohexamide (CAS 968-81-0)
QUICK LINKS
Acetohexamide functions as a hypoglycemic agent in experiment. It exerts its mechanism of action by stimulating the release of insulin from pancreatic beta cells, thereby lowering blood glucose levels. Acetohexamide targets the sulfonylurea receptor on the pancreatic beta cells, leading to the closure of ATP-sensitive potassium channels. As a result, the depolarization of the cell membrane occurs, leading to an influx of calcium ions and subsequent release of insulin. Acetohexamide′s role in experiment involves its ability to modulate insulin secretion through its interaction with specific cellular receptors and channels. Acetohexamide′s mechanism of action at the molecular level involves its specific binding to the sulfonylurea receptor and subsequent effects on potassium channels and insulin release.
Acetohexamide (CAS 968-81-0) References
- Acetohexamide-acetazolamide mix-up during emergency treatment. | Sabb, PC. and Allen, JC. 1999. Am J Health Syst Pharm. 56: 2463. PMID: 10595808
- Acetohexamide: comparison with other sulfonylurea compounds in the treatment of diabetes mellitus. | BOSHELL, BR., et al. 1962. Clin Pharmacol Ther. 3: 750-7. PMID: 14014164
- ACETOHEXAMIDE IN TREATMENT OF DIABETES MELLITUS. | MONTGOMERY, DA., et al. 1964. Br Med J. 1: 868-71. PMID: 14104973
- Chromatographic analysis of acetohexamide binding to glycated human serum albumin. | Joseph, KS., et al. 2010. J Chromatogr B Analyt Technol Biomed Life Sci. 878: 2775-81. PMID: 20829128
- Metabolic reduction of acetohexamide in rat kidney: sex difference and effect of streptozotocin-induced diabetes. | Kojima, Y., et al. 1988. J Pharmacobiodyn. 11: 309-13. PMID: 2971793
- Rabbit dehydrogenase/reductase SDR family member 11 (DHRS11): Its identity with acetohexamide reductase with broad substrate specificity and inhibitor sensitivity, different from human DHRS11. | Endo, S., et al. 2019. Chem Biol Interact. 305: 12-20. PMID: 30926317
- In Vitro Investigations of Acetohexamide Binding to Glycated Serum Albumin in the Presence of Fatty Acid. | Szkudlarek, A., et al. 2020. Molecules. 25: PMID: 32429512
- Potential Small Molecules for Therapy of Lupus Nephritis Based on Genetic Effect and Immune Infiltration. | Qing, J., et al. 2022. Biomed Res Int. 2022: 2259164. PMID: 35502341
- Acetohexamide in the therapy of diabetes mellitus. | Balodimos, MC., et al. 1968. Metabolism. 17: 669-80. PMID: 5676213
- Carbonyl reductase activity for acetohexamide in human erythrocytes. | Kishimoto, M., et al. 1994. Drug Metab Dispos. 22: 367-70. PMID: 8070312
- Substitution of acetohexamide for acetazolamide. | Rutzen, AR. and Weiss, JS. 1993. Am J Ophthalmol. 116: 379-80. PMID: 8357069
- Inadvertent substitution of acetohexamide for acetozolamide. | Hargett, NA., et al. 1977. Am J Ophthalmol. 84: 580-3. PMID: 910864
- Characterization of acetohexamide reductases purified from rabbit liver, kidney, and heart: structural requirements for substrates and inhibitors. | Imamura, Y., et al. 1997. J Biochem. 121: 705-10. PMID: 9163521
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Acetohexamide, 1 g | sc-254935 | 1 g | $52.00 |
