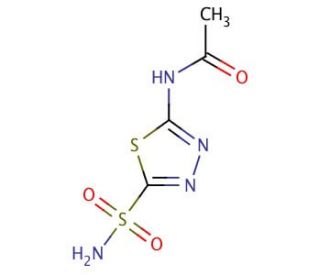
Molecular structure of Acetazolamide, CAS Number: 59-66-5
Acetazolamide (CAS 59-66-5)
See product citations (1)
Alternate Names:
5-Acetamido-1,3,4-thiadiazole-2-sulfonamide; Acetamox; Atenezol
Application:
Acetazolamide is a carbonic anhydrase inhibitor
CAS Number:
59-66-5
Purity:
≥98%
Molecular Weight:
222.25
Molecular Formula:
C4H6N4O3S2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Acetazolamide is a potent inhibitor of the enzyme carbonic anhydrase, which is involved in the regulation of pH balance and bicarbonate levels in the body. Acetazolamide inhibits the enzyme by binding to its active site and preventing the enzyme from catalyzing the reaction of carbon dioxide and water to form carbonic acid. This inhibition leads to a decrease in the production of bicarbonate, which in turn leads to a decrease in the pH of the body fluids. It also inhibits cell membrane water permeability via interaction with aquaporins.
Acetazolamide (CAS 59-66-5) References
- Acetazolamide treatment for infantile central sleep apnea. | Philippi, H., et al. 2001. J Child Neurol. 16: 600-3. PMID: 11510934
- Acetazolamide: future perspective in topical glaucoma therapeutics. | Kaur, IP., et al. 2002. Int J Pharm. 248: 1-14. PMID: 12429455
- Acetazolamide: a forgotten diuretic agent. | Kassamali, R. and Sica, DA. 2011. Cardiol Rev. 19: 276-8. PMID: 21983315
- Acetazolamide for the treatment of idiopathic intracranial hypertension. | Supuran, CT. 2015. Expert Rev Neurother. 15: 851-6. PMID: 26154918
- Severe ocular side effects with acetazolamide: case report. | Llovet-Rausell, A., et al. 2016. Arch Soc Esp Oftalmol. 91: 543-546. PMID: 27179669
- Evaluating off-label uses of acetazolamide. | Van Berkel, MA. and Elefritz, JL. 2018. Am J Health Syst Pharm. 75: 524-531. PMID: 29626002
- Paradoxical Critical Hyperkalemia After Acetazolamide for Cerebrovascular Reactivity Study: A Case Report. | Burbridge, MA. and Jaffe, RA. 2020. A A Pract. 14: 69-71. PMID: 31770135
- Influence of Acetazolamide on Hand Strength and Manual Dexterity During a 30-h Simulated High Altitude Exposure. | Yurkevicius, BR., et al. 2020. Mil Med. 185: e1161-e1167. PMID: 32175586
- Optimization of Acetazolamide-Based Scaffold as Potent Inhibitors of Vancomycin-Resistant Enterococcus. | Kaur, J., et al. 2020. J Med Chem. 63: 9540-9562. PMID: 32787141
- Acetazolamide responsive hereditary paroxysmal ataxia. | Friedman, JH. and Hollmann, PA. 1987. Mov Disord. 2: 67-72. PMID: 3332806
- Oral acetazolamide for intraocular pressure lowering: balancing efficacy and safety in ophthalmic practice. | Gulati, S. and Aref, AA. 2021. Expert Rev Clin Pharmacol. 14: 955-961. PMID: 34003717
- Acetazolamide modulates intracranial pressure directly by its action on the cerebrospinal fluid secretion apparatus. | Barbuskaite, D., et al. 2022. Fluids Barriers CNS. 19: 53. PMID: 35768824
- Heart failure: an update from the last years and a look at the near future. | Riccardi, M., et al. 2022. ESC Heart Fail. 9: 3667-3693. PMID: 36546712
- Determination of acetazolamide in biological fluids by high-performance liquid chromatography. | Gal, J., et al. 1981. Curr Eye Res. 1: 361-5. PMID: 7307542
Inhibitor of:
4833422F24Rik, 4930455F23Rik, ACPL2, ACY1L2, AE3, Aldolase C, AQP1, AQP12, AQP12A, AQP12B, AQP3, AQP4, AQP6, C20orf111, C4ST-2, CA I, CA III, CA IV, CA VI, CA VII, CA XII, CA XIII, CA XV, Carbonic Anhydrases, CLCA3_Clac3, Dnajc22, Enzyme, ERMAP, FIBCD1, Gastrokine 1, Gastrokine 3, GlcNAc kinase, HRASLS, LOC143666, LOXL4, NAG, NBC4, NCBE, Olfactomedin (OLFM), Olfr666, Olfr740, Olfr741, Olfr742, Olfr744, Olfr745, Olfr746, Olfr824, Olr252, OTOP2, PIPES, PNKD, Proliferin-3, retbindin, RhBG, SCO2, SLC10A5, SLC26A11, SLC45A4, SLC46A3, SLC4A (Solute carrier family 4), SLC4A7, UAP1L1, V1RD1, and Xg.Activator of:
AQP12A, Barttin, CA XIV, CLIC6, Hemoglobin μ, KCC2b, LOC622307, NHE-5, NHE-7, NPT1, Pma2, Prestin, SLC7A14, Sodium Chloride, and TCIRG1.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Acetazolamide, 10 g | sc-214461 | 10 g | $81.00 | |||
Acetazolamide, 25 g | sc-214461A | 25 g | $177.00 | |||
Acetazolamide, 100 g | sc-214461B | 100 g | $434.00 | |||
Acetazolamide, 250 g | sc-214461C | 250 g | $541.00 | |||
Acetazolamide, 500 g | sc-214461D | 500 g | $883.00 | |||
Acetazolamide, 1 kg | sc-214461E | 1 kg | $1479.00 | |||
Acetazolamide, 2 kg | sc-214461F | 2 kg | $2244.00 |
