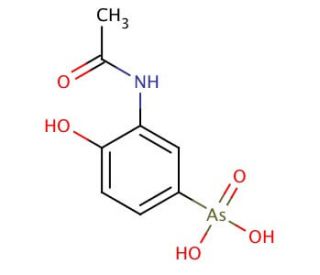
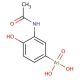
Molecular structure of Acetarsone, CAS Number: 97-44-9
Acetarsone (CAS 97-44-9)
Alternate Names:
N-Acetyl-4-hydroxy-m-arsanilic acid; Acetarsol
Application:
Acetarsone is an antiifective agent used as an antiprotozoal
CAS Number:
97-44-9
Molecular Weight:
275.09
Molecular Formula:
C8H10AsNO5
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Acetarsone is an organoarsenic compound. This allows the study of the chemical properties and reactivities associated with arsenic in an organic framework. Acetarsone may bind to protein-containing sulfhydryl groups forming As-S bonds. It is a member of acetamides and anilides. Acetarsone is the acetyl structural analog of 3-amino-4-hydroxy-phenyl-arsonic acid. According to one study, Its ionization constants are pK1 = 3.73, pK2 = 7.9, and pK3 = 9.3, with the second ionization step attributed to the phenolic proton. Acetarsone can stimulate cell proliferation at micromolar levels. It has the ability to permeate through intestinal epithelial cells.
Acetarsone (CAS 97-44-9) References
- Acute systemic reaction to acetarsol. | WHITE, A. 1956. Br Med J. 2: 1528-9. PMID: 13374373
- A survey of taeniid cestodes in farm dogs in mid-Wales. | Jones, A. and Walters, TM. 1992. Ann Trop Med Parasitol. 86: 137-42. PMID: 1417204
- Syphilis and intravaginal treatment of trichomoniasis with compounds containing acetarsone. | PUTKONEN, T. 1949. Ann Med Exp Biol Fenn. 27: 137-44. PMID: 15392081
- The use of acetarsone, carbarsone, and iodochlorohydroxyquinoline. | GREEN, MW. 1946. Bull Am Pharm Assoc. 14: 75-80. PMID: 21023231
- Surface-enhanced Raman scattering (SERS) characterization of trace organoarsenic antimicrobials using silver/polydimethylsiloxane nanocomposites. | Olavarría-Fullerton, J., et al. 2011. Appl Spectrosc. 65: 423-8. PMID: 21396190
- Speciation of arsenic in urine following intravenous administration of arsthinol in mice. | Ajana, I., et al. 2010. Eur J Drug Metab Pharmacokinet. 35: 59-65. PMID: 21495268
- Permeation of roxarsone and its metabolites increases caco-2 cell proliferation. | Bayse, GS., et al. 2013. Adv Biol Chem. 3: 389-396. PMID: 25632371
- Acetarsol in the management of mesalazine-refractory ulcerative proctitis: a tertiary-level care experience. | Argyriou, K., et al. 2019. Eur J Gastroenterol Hepatol. 31: 183-186. PMID: 30499784
- [Acetarsol-an organic arsenic compound with toxic effect on the optic nerve]. | Berggren, L. 1988. Lakartidningen. 85: 521-3. PMID: 3352424
- Ultraviolet spectrum correlations with the conjugate acid-base species of acetarsone and arsthinol. | Hiskey, CF. and Cantwell, FF. 1968. J Pharm Sci. 57: 2105-11. PMID: 5708352
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Acetarsone, 50 g | sc-227187 | 50 g | $122.00 |
