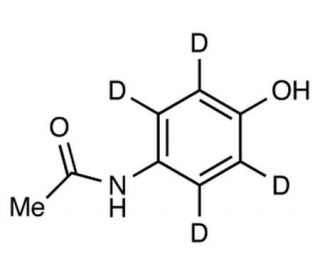
Acetaminophen-D4 (Major) (CAS 64315-36-2)
See product citations (5)
QUICK LINKS
Acetaminophen-D4 is a deuterated form of acetaminophen may be used analgesic and antipyretic agent. It functions as an inhibitor of the enzyme cyclooxygenase, particularly the COX-2 isoform, leading to a reduction in the production of prostaglandins. This inhibition results in a decrease in pain and fever, which may be useful for studying the role of prostaglandins in various biological processes. Acetaminophen-D4 is also known to act on the central nervous system, exerting its effects through modulation of the endocannabinoid system. Acetaminophen-D4′s mechanism of action may involve the activation of cannabinoid receptors, leading to the modulation of pain perception and temperature regulation. In experimental applications, Acetaminophen-D4 is utilized to investigate the specific molecular pathways involved in pain and fever regulation, providing insights into the underlying mechanisms of these physiological processes. Its deuterated form allows for precise tracking and quantification in pharmacokinetic and metabolic studies, contributing to a deeper understanding of its pharmacological properties.
Acetaminophen-D4 (Major) (CAS 64315-36-2) References
- Direct injection mass spectrometric confirmation of multiple drugs in overdose cases from postmortem blood using electrospray ionization-tandem mass spectrometry and MS(3). | Peer, CJ., et al. 2008. J Anal Toxicol. 32: 709-14. PMID: 19007526
- Evaluating human liver reserve function by measuring serum concentrations of phenacetin and its metabolites. | Xiong, WJ., et al. 2010. J Dig Dis. 11: 358-63. PMID: 21091898
- Acetaminophen, pesticide, and diethylhexyl phthalate metabolites, anandamide, and fatty acids in deciduous molars: potential biomarkers of perinatal exposure. | Camann, DE., et al. 2013. J Expo Sci Environ Epidemiol. 23: 190-6. PMID: 22805989
- Comparison of the quantification of acetaminophen in plasma, cerebrospinal fluid and dried blood spots using high-performance liquid chromatography-tandem mass spectrometry. | Taylor, RR., et al. 2013. J Pharm Biomed Anal. 83: 1-9. PMID: 23670126
- Acetaminophen metabolism after liver resection: A prospective case-control study. | Hughes, MJ., et al. 2015. Dig Liver Dis. 47: 1039-46. PMID: 26362614
- Metabolism of AM404 From Acetaminophen at Human Therapeutic Dosages in the Rat Brain. | Muramatsu, S., et al. 2016. Anesth Pain Med. 6: e32873. PMID: 27110534
- Multi-residue determination of micropollutants in Phragmites australis from constructed wetlands using microwave assisted extraction and ultra-high-performance liquid chromatography tandem mass spectrometry. | Petrie, B., et al. 2017. Anal Chim Acta. 959: 91-101. PMID: 28159109
- Bioavailability of suppository acetaminophen in healthy and hospitalized ill dogs. | Sikina, ER., et al. 2018. J Vet Pharmacol Ther. 41: 652-658. PMID: 29756216
- Simultaneous determination of acetaminophen and oxycodone in human plasma by LC-MS/MS and its application to a pharmacokinetic study. | Lu, W., et al. 2018. J Pharm Anal. 8: 160-167. PMID: 29922484
- Development and characterization of rat duodenal organoids for ADME and toxicology applications. | Hedrich, WD., et al. 2020. Toxicology. 446: 152614. PMID: 33199268
- Intravenous Acetaminophen Does Not Provide Adequate Postoperative Analgesia in Dogs Following Ovariohysterectomy. | Leung, J., et al. 2021. Animals (Basel). 11: PMID: 34944384
- Acetaminophen production in man after coadministration of acetanilid and phenacetin. A study with stable isotopes. | Baty, JD. and Robinson, PR. 1977. Clin Pharmacol Ther. 21: 177-86. PMID: 837636
- Quantitative LC/MS/MS analysis of acetaminophen–cysteine adducts (APAP–CYS) and its application in acetaminophen overdose patients | T. Hairin,a A. R. Marzilawati,b E. M. H. Didi,a S. Mahadeva,b Y. K. Lee,c N. Abd. Rahman,c A. M. Mustafaa and Z. Chik*a. 2013. Anal. Methods,.,5,: 1955-1964.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Acetaminophen-D4 (Major), 10 mg | sc-217563 | 10 mg | $312.00 |
