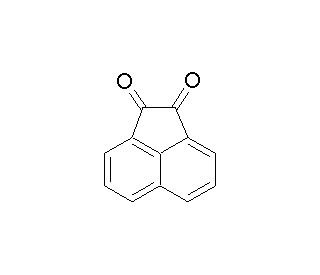
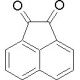
Molecular structure of Acenaphthenequinone, CAS Number: 82-86-0
Acenaphthenequinone (CAS 82-86-0)
Application:
Acenaphthenequinone is a substituted naphthalene compound
CAS Number:
82-86-0
Purity:
95%
Molecular Weight:
182.17
Molecular Formula:
C12H6O2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Acenaphthenequinone is a quinoid polycyclic aromatic hydrocarbon, which acts as a redox-active compound. Studies suggest that Acenaphthenequinone can produce reactive oxygen species (ROS) that result in damage to A549 Cell Lysate (sc-2413). It is known that most polycyclic aromatic hydrocarbons are contained in diesel exhaust particles (DEPs) in the air. Since these species are damaging to human lung tissues, Acenaphthenequinone is essential in the study of this process. In addition, Acenaphthenequinone induces the expression of Cox-2 and translocates NF-κB from the cytosol to the nucleus during the synthesis of reactive oxygen species.
Acenaphthenequinone (CAS 82-86-0) References
- An environmental quinoid polycyclic aromatic hydrocarbon, acenaphthenequinone, modulates cyclooxygenase-2 expression through reactive oxygen species generation and nuclear factor kappa B activation in A549 cells. | Chung, SW., et al. 2007. Toxicol Sci. 95: 348-55. PMID: 17082565
- Activation of 5-lipoxygenase and NF-kappa B in the action of acenaphthenequinone by modulation of oxidative stress. | Chung, SW., et al. 2008. Toxicol Sci. 101: 152-8. PMID: 17925309
- Functional, Aromatic, and Fluorinated Monothiosemicarbazones: Investigations into Their Structures and Activity toward the Gallium-68 Incorporation by Microwave Irradiation. | Sarpaki, S., et al. 2022. ACS Omega. 7: 13750-13777. PMID: 35559172
- Methylene Blue as a Photo-Redox Catalyst: The Development Synthesis of Tetrahydrobenzo[b]pyran Scaffolds via a Single-Electron Transfer/Energy Transfer. | Mohamadpour, F. 2022. Front Chem. 10: 934781. PMID: 35923256
- Ruthenium Complexes Bearing α-Diimine Ligands and Their Catalytic Applications in N-Alkylation of Amines, α-Alkylation of Ketones, and β-Alkylation of Secondary Alcohols. | Gayathri, S., et al. 2022. ACS Omega. 7: 33107-33122. PMID: 36157732
- Acridine yellow G (AYG) as a photo-induced electron transfer (PET) photocatalyst employed for the radical Michael-Mannich cyclocondensation of imines. | Mohamadpour, F. 2022. Front Chem. 10: 1015330. PMID: 36300032
- Visible-light-driven radical Friedländer hetero-annulation of 2-aminoaryl ketone and α-methylene carbonyl compound via organic dye fluorescein through a single-electron transfer (SET) pathway. | Mohamadpour, F. 2022. BMC Chem. 16: 116. PMID: 36522648
- Photosensitive damage of dipeptides: mechanism and influence of structure. | Yang, C., et al. 2023. Phys Chem Chem Phys. 25: 4923-4928. PMID: 36722384
- Structural and Functional Diversity in Rigid Thiosemicarbazones with Extended Aromatic Frameworks: Microwave-Assisted Synthesis and Structural Investigations. | Cortezon-Tamarit, F., et al. 2023. ACS Omega. 8: 16047-16079. PMID: 37179648
- Cobalt(II) coordination to an N4-acenaphthene-based ligand and its sodium complex. | Khrizanforova, VV., et al. 2023. Dalton Trans. 52: 7876-7884. PMID: 37212425
- A specific dispiropiperazine derivative that arrests cell cycle, induces apoptosis, necrosis and DNA damage. | Liu, VP., et al. 2023. Sci Rep. 13: 8674. PMID: 37248333
- Structure-property relationships in dicyanopyrazinoquinoxalines and their hydrogen-bonding-capable dihydropyrazinoquinoxalinedione derivatives. | Akhmedov, TN., et al. 2024. Beilstein J Org Chem. 20: 1037-1052. PMID: 38746652
- Various synthesis and biological evaluation of some tri -tetra-substituted imidazoles derivatives: A review. | Hamdi, A., et al. 2024. Heliyon. 10: e31253. PMID: 38803909
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Acenaphthenequinone, 25 g | sc-239180 | 25 g | $55.00 |
