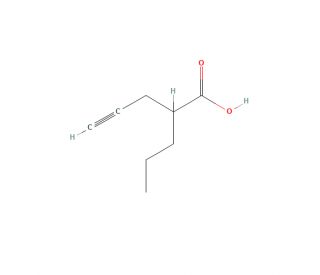
Molecular structure of ABS 205, CAS Number: 24102-11-2
ABS 205 (CAS 24102-11-2)
See product citations (1)
Alternate Names:
4-yn-VPA
Application:
ABS 205 is a valproic acid derivative
CAS Number:
24102-11-2
Molecular Weight:
140.18
Molecular Formula:
C8H12O2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Orally active valproic acid derivative that exhibits neuroprotective and teratogenic activity. Upregulates neural cell adhesion molecule (NCAM) expression, activates PPARδ (IC50 = 0.6 mM) and increases Hoxa1 expression in rat embryos. Antiproliferative; induces G1 cell cycle arrest in C6 glioma cells (IC50 ~ 2 mM).
ABS 205 (CAS 24102-11-2) References
- Modulation of peroxisome proliferator-activated receptor delta activity affects neural cell adhesion molecule and polysialyltransferase ST8SiaIV induction by teratogenic valproic acid analogs in F9 cell differentiation. | Lampen, A., et al. 2005. Mol Pharmacol. 68: 193-203. PMID: 15829700
- S-2-pentyl-4-pentynoic hydroxamic acid and its metabolite s-2-pentyl-4-pentynoic acid in the NMRI-exencephaly-mouse model: pharmacokinetic profiles, teratogenic effects, and histone deacetylase inhibition abilities of further valproic acid hydroxamates and amides. | Eikel, D., et al. 2006. Drug Metab Dispos. 34: 612-20. PMID: 16415118
- Induction of the homeotic gene Hoxa1 through valproic acid's teratogenic mechanism of action. | Stodgell, CJ., et al. 2006. Neurotoxicol Teratol. 28: 617-24. PMID: 16989981
- On the development of alternative antiepileptic drugs. Lack of enantioselectivity of the anticonvulsant activity, in contrast to teratogenicity, of 2-n-propyl-4-pentenoic acid and 2-n-propyl-4-pentynoic acid, analogues of the anticonvulsant drug valproic acid. | Hauck, RS., et al. 1991. Naturwissenschaften. 78: 272-4. PMID: 1922389
- Cell proliferation, migration and CAM-dependent neurite outgrowth as developmental in vitro endpoints for screening teratogenic potential: Application to valproic acid and related analogues of varying potency. | Bacon, CL., et al. 1998. Toxicol In Vitro. 12: 101-9. PMID: 20654391
- Stereoselective dysmorphogenicity of the enantiomers of the valproic acid analogue 2-N-propyl-4-pentynoic acid (4-yn-VPA): cross-species evaluation in whole embryo culture. | Andrews, JE., et al. 1997. Teratology. 55: 314-8. PMID: 9261925
- The enantioselective teratogenicity of 2-n-propyl-4-pentynoic acid (4-yn-VPA) is due to stereoselective intrinsic activity and not differences in pharmacokinetics | R.-S. Hauck, M.M.A. Elmazar 1, C. Plum, H. Nau. 1992. Toxicology Letters. 60: 145-153.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
ABS 205, 10 mg | sc-203797 | 10 mg | $200.00 | |||
ABS 205, 50 mg | sc-203797A | 50 mg | $525.00 |
