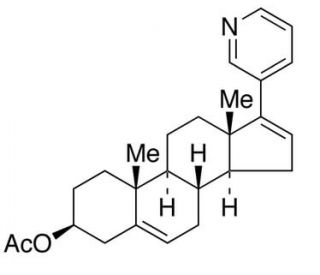
Molecular structure of Abiraterone Acetate, CAS Number: 154229-18-2
Abiraterone Acetate (CAS 154229-18-2)
See product citations (1)
Alternate Names:
(3β)-17-(3-Pyridinyl)androsta-5,16-dien-3-ol Acetate
Application:
Abiraterone Acetate is a steroidal inhibitor of CYP17A1 in androgen biosynthesis
CAS Number:
154229-18-2
Molecular Weight:
391.55
Molecular Formula:
C26H33NO2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Abiraterone acetate has been seen to be a novel steroidal inhibitor of CYP17A1(C17,20 lyase), a rate-limiting enzyme in androgen biosynthesis. The compound has been shown to be apporximately 21-24 times more potent than ketoconazole (sc-200496) and liarozole (sc-204055) in CYP17A1 lyase inhibition. Abiraterone Acteate is actively being researched as a treatment for castration resistent prostate cancer.
Abiraterone Acetate (CAS 154229-18-2) References
- Hormonal impact of the 17alpha-hydroxylase/C(17,20)-lyase inhibitor abiraterone acetate (CB7630) in patients with prostate cancer. | O'Donnell, A., et al. 2004. Br J Cancer. 90: 2317-25. PMID: 15150570
- Are we meeting their needs. | McDonnell, E. 1992. Br J Theatre Nurs. 2: S7-12. PMID: 1627848
- ThermoTRP channels as modular proteins with allosteric gating. | Latorre, R., et al. 2007. Cell Calcium. 42: 427-38. PMID: 17499848
- CYP17 blockade by abiraterone: further evidence for frequent continued hormone-dependence in castration-resistant prostate cancer. | Ang, JE., et al. 2009. Br J Cancer. 100: 671-5. PMID: 19223900
- Steroid profiling in H295R cells to identify chemicals potentially disrupting the production of adrenal steroids. | Strajhar, P., et al. 2017. Toxicology. 381: 51-63. PMID: 28235592
- Zoledronic Acid Prevents Bone Resorption Caused by the Combination of Radium-223, Abiraterone Acetate, and Prednisone in an Intratibial Prostate Cancer Mouse Model. | Suominen, MI., et al. 2023. Cancers (Basel). 15: PMID: 37627143
- Novel steroidal inhibitors of human cytochrome P45017 alpha (17 alpha-hydroxylase-C17,20-lyase): potential agents for the treatment of prostatic cancer. | Potter, GA., et al. 1995. J Med Chem. 38: 2463-71. PMID: 7608911
- Pharmacology of novel steroidal inhibitors of cytochrome P450(17) alpha (17 alpha-hydroxylase/C17-20 lyase). | Barrie, SE., et al. 1994. J Steroid Biochem Mol Biol. 50: 267-73. PMID: 7918112
- Overview of phase III trials on combined androgen treatment in patients with metastatic prostate cancer. | Denis, L. and Murphy, GP. 1993. Cancer. 72: 3888-95. PMID: 8252511
- Novel nonsteroidal inhibitor of cytochrome P450(17alpha) (17alpha-hydroxylase/C17-20 lyase), YM116, decreased prostatic weights by reducing serum concentrations of testosterone and adrenal androgens in rats. | Ideyama, Y., et al. 1998. Prostate. 37: 10-8. PMID: 9721064
- Solubility Measurements at 296 and 310 K and Physicochemical Characterization of Abiraterone and Abiraterone Acetate | Solymosi, T., Tóth, F., Orosz, J., Basa-Dénes, O., Angi, R., Jordán, T.,.. & Glavinas, H. 2018. Journal of Chemical & Engineering Data. 63(12): 4453-4458.
Inhibitor of:
17β-HSD6, 3β-HSD4, 3β-HSD5, 3β-HSD7, 5 alpha-Reductase 2, AKR1D1, ARL13B, ARL3, Choriogonadotropin β, Choriogonadotropin Holo c3, Cortisol, CYP11A1, CYP11B, CYP17A1, CYP26C1, EG434676, EG545948, EG623898, EG626305, EG639025, INSL3, Kap, MISR II, NURIM, OTTMUSG00000008394, PGCP, Pma, PSGR, SMR2, TSARG6, Tsx, and Vmn2r23.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Abiraterone Acetate, 5 mg | sc-207240 | 5 mg | $231.00 |
